
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
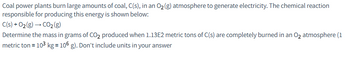
Transcribed Image Text:Coal power plants burn large amounts of coal, C(s), in an O₂(g) atmosphere to generate electricity. The chemical reaction
responsible for producing this energy is shown below:
C(s) +0₂(g) → CO₂ (g)
Determine the mass in grams of CO₂ produced when 1.13E2 metric tons of C(s) are completely burned in an O₂ atmosphere (1
metric ton = 10³ kg = 106 g). Don't include units in your answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A household water heater containing 50 gallons (227 L) of water is heated burning natural gas (methane, molar mass = 16.043 g mol-1) via the following reaction: CH4(g) + O2(g) CO2(g) + H20(O A,H = -891 kJ mol-1 What mass of methane, in units of g, is needed to heat the 227 L of water in the heater from 25.0°C to 44.0°C? Assume: • the heater is 100% efficient in its use of heat energy • no heat is lost to the surroundings specific heat of water is: 4.184 Jg-1 °C-1 density of water is: 0.9970 g mL¯1arrow_forwardNitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. In the first step, nitrogen and hydrogen react to form ammonia: N,(0) + 3 H,(9) → 2 NH,(9) ΔΗ-92. k In the second step, ammonia and oxygen react to form nitric acid and water: NH,(9) + 20,(9) → HNO3(g) + H,O(g) AH=-330. kJ Calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. Round your answer to the nearest kJ. ?arrow_forwardBalance the following equation. What is the sum of the coefficients in the balanced equation? KCIO;(s) + P(s) - P,O10(s)+ KCI(s)arrow_forward
- 2arrow_forward3. Determine the enthalpy change (AH₂) for the final reaction below. a. CO(g) + ½ O₂(g) → CO₂(g) AH, -67.6 kcal b. N₂(g) + O₂(g) →→→ 2NO(g) AH₂ = 43.2 kcal c. CO(g) + NO(g) →→→ CO₂(g) + ¹½ N₂(g) AH₂ = ?arrow_forwardWhen heated, metal hydroxides decompose to produce a metal oxide and water. Selected the correct balanced equation for the decomposition of calcium hydroxide. CaOH (s) → CaO2 (s) + H2O (g) Ca(OH)2 (s) → CaO (s) + H2O (g) 2 CaOH (s) → 2 CaO (s) + H2O (g) 3 Ca(OH)2 (s) → 3 CaO2 (s) + H2O (g)arrow_forward
- Ethanol, C,H,O, is most often blended with gasoline - usually as a 10 percent mix - to create a fuel called gasohol. Ethanol is a renewable resource and ethanol-blended fuels, like gasohol, appear to burn more efficiently in combustion engines. The heat of combustion of ethanol is 326.7 kcal/mol. The heat of combustion of 2-methylheptane, C3H18, is 1.306×10³ kcal/mol. How much energy is released during the complete combustion of 482 grams of 2-methylheptane ? kcal Assuming the same efficiency, would 482 grams of ethanol provide more, less, or the same amount of energy as 482 grams of 2- methylheptane v more less the same amountarrow_forwardBased on the balance reaction between iron and oxygen, how many electrons are transferred when 27 g of iron react? balanced reaction: 4Fe(s)+3O2(g)⟶2Fe2O3(s)arrow_forwardYour lab partner accidentally mixed some sodium chloride with your sample of Epsom salts (MgSO₄・7H₂O). You want to make a standard solution of magnesium ion, and this is the only sample of a magnesium salt you have. To determine the amount of magnesium salt in the mixture, you heat 100.00 g to drive off the water of hydration and find that the anhydrous mixture has a mass of 57.75 g. How many grams of the original salt mixture must you add to 1.000 L of water to make a 0.100 M solution of Mg²⁺ ion?arrow_forward
- The enthalpy change of neutralization is -55.8 kJ/mol for a strong base strong acid reaction generating water. If you have 0.5 mol of H2SO4 and 0.5 mol of NaOH, what is the balanced chemical reaction, and calculate the limiting reagent for the reaction?arrow_forward01 04 in the balanced equation for the complete combustion of heptane, the coefficient for carbon dioxide is: Cu + O = CO + HO Warrow_forwardThe following unbalanced equation illustrates the overall reaction by which the body utilizes glucose to produce energy: C6H12O6(s) + O2(g) → CO2(g) + H2O(l) What is the conversion factor that allows for the calculation of moles of carbon dioxide based on moles of glucose?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY