
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
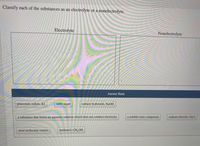
Transcribed Image Text:Classify each of the substances as an electrolyte or a nonelectrolyte.
Electrolyte
Nonelectrolyte
Answer Bank
potassium iodide, KI
table sugar
sodium hydroxide, NaOH
a substance that forms an aqueous solution which does not conduct electricity
a soluble ionic compound
sodium chloride, NaCl
most molecular solutes
methanol, CH,OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Suppose you have a bottle of concentrated sulfuric acid (18.0 M). How would you prepare 250 mL of a 3.0 M solution of sulfuric acid? Show step by step.arrow_forward12arrow_forwardCalculate the molarity of Cl− anions in the chemist's solution. Be sure your answer is rounded to 2 significant digits.arrow_forward
- How do I write the total ionic equation and the net ionic equation?Nitric acid and silver chloridearrow_forwardFill in the blanks by doing the necessary calculations with the information provided. a) pH = 12.3 [H+] = _______________ b) [OH-] = 2.29 x 10-11 [H+] = __________ c) [H+] = 4.07 x 10-7 pH = __________ d) [H+] = 9.93 x 10-4 pOH = ____________arrow_forwardWrite and explain step by step. If you are handwriting it make sure its readible.arrow_forward
- Consider a solution prepared by combining 45.0 g of BaCl2 with enough water to prepare a 750.0 mL of solution. Assume that the volume of water added was 748 mL. Density of water is 1.00 g/mL. 1) What volume of this solution would be required to prepare 500.0 mL of a solution that is 0.0034 M in BaCl2?arrow_forwardSodium hydroxide is a substance that causes severe burns. It is used in many educational laboratory activities, but because of its hazardous nature, care must be taken to ensure its safe use in the lab. Select the statements that are true regarding the nature of sodium hydroxide and the safety precautions that should be used when handling it in the laboratory. Sodium hydroxide solutions with high concentration can cause severe burns. If sodium hydroxide is spilled on skin or clothing, the contaminated clothing should be removed immediately and the affected area should be drenched with plenty of water. Medical attention should be sought if a large area is affected, or if blistering occurs. Eye protection should be worn at all times when handling any form of sodium hydroxide. Sodium hydroxide solutions are particularly dangerous to the eyes. If sodium hydroxide is introduced to the eyes, flush the affected eye continuously with water for at least 15 min. Get medical attention…arrow_forwardWhich of the following solutes would form electrolyte solutions when dissolved in water? Choose all that are correct. Group of answer choices sulfuric acid, H2SO4 ethanol, C2H5OH sodium acetate, NaC2H3O2 ammonium carbonate, (NH4)2CO3 fructose, C6H12O6 carbon dioxide, CO2arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY