
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Calculate the percent error between your average experimental value and theoretical value for the mass percent
of oxygen in KClO3. Show your work clearly.
ev = 40.767%
tv = 39.2%
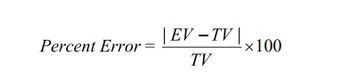
Transcribed Image Text:Percent Error:
| EV - TV |
TV
×100
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- You analyzed 2.475 g of a mixture of NaCl, SiO2, and CaCO3. You isolate 0.825 g NaCl, 0.825 g of CaCO3, and 0.580 g SiO2. You were told that the original mixture contained 35% NaCl, 45% CaCO3, and 20% SiO2. Report two possible sources of error.arrow_forwardThe formula of a compound is X20. Which of the following is X least likely to be? a. Barium b. Sodium c. Cesium d. Hydrogenarrow_forwardIs this correct answer 52.144%Please show me the work with this correct answer 52.144%arrow_forward
- Can I get help with the following question pleasearrow_forwardn lo Report Form, Density of Nails, Baby Oll, ana MOPE Analysis of Results and Other Questions Densities (in g/cm at 25°C) 6. Assuming your nails are made of one of the elemental metals in the table (right), what is the most likely Mg 1.74 metal? How confident are you that they are made up of that metal? Explain. Zn 7.14 Cr 7.15 Sn 7.26 niloded lo sl Fe 7.87 Ni 8.90 8.96 Pb 11.3 Au 19.3 o dee 7. (a) Search the web for a value for the density of baby oil (if no T is given, it is assumed to be near room T): Source company or site name where you got the value (don't list the full URL): (b) Search the web (Google is best here) for the density of mineral oil from Sigma-Aldrich (a chemical company): (c) As you learned during your prelab (hopefully!), baby oil and mineral oil are not pure substances. As such, their composition (and thus density) can vary a bit, which would not be so for a pure substance (at a given T). That is why you likely found varying values-or ranges of values-for baby…arrow_forward(2a-302-62.73) Which of the following is the lightest? Show all work to explain your answer. 15.8 mol of helium 3.11 x 1023 molecules of nitrogen trichloride 62.73 g of titaniumarrow_forward
- Find the mass of 2.45 X 1023 molecules of Ca(OH)2? Use: Ca = 42.0g, H = 1.25g and O = 15.7g as average atomic mass.arrow_forwardWhat is the average atomic weight for a hypothetical element "Q" which is composed of the following mass isotopes and their corresponding percent mass abundances? Hint: express your answer to four significant figures or risk losing all credit! Do not include the unit "amu" in your answer. mass of % mass isotope abundance (amu) 39.63 32.11 40.59 55.24 42.99 12.65arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY