
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
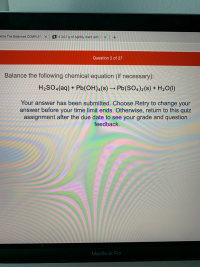
Transcribed Image Text:Balance the following chemical equation (if necessary):
H2SO«(aq) + Pb(OH)4(s) → Pb(SO4)2(s) + H2O(I)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In sediments and waterlogged soil, dissolved O2 concentrations are so low that the microorganisms living there must rely on other sources of oxygen for respiration. Some bacteria can extract the oxygen in sulfate ions, reducing the sulfur in them to hydrogen sulfide gas and giving the sediments or soil a distinctive rotten-egg odor.Write the net ionic equation for the reaction under acidic conditions (H3O+) that releases O2 from sulfate and forms hydrogen sulfide gas. Use water as the reactant in the half-reaction that describes the formation of oxygen.arrow_forwardZinc metal reacts with nitric acid, HNO, to produce a number of products, depending on how dilute the acid solution is. In a concentrated salution, zinc reduces nitrate ion to ammonium ion; zinc is oxidized to zinc ion, Zn Write the net ionic equation for this reaction.arrow_forwardBe sure to answer all parts. Consider the following balanced redox reaction (do not include state of matter in your answers): 2CrO₂ (aq) + 2H₂O()+6C10 (aq) → 2CrO42 (aq) + 3Cl₂(g) + 40H (aq) (a) Which species is being oxidized? (b) Which species is being reduced? (c) Which species is the oxidizing agent? (d) Which species is the reducing agent? (e) From which species to which does electron transfer occur? Electrons transfer from toarrow_forward
- 1. a) Write the formula for the conjugate base of HPO4^-2 b) Write the formula for the conjugate acid of NH3arrow_forwardComplete the balanced neutralization equation for the reaction below: H₂SO₄(aq)+Ca(OH)₂(aq)→arrow_forwardComplete the balanced neutralization equation for the reaction below: HC₂H₃O₂(aq)+Sr(OH)₂(aq)→arrow_forward
- Chemists working with fluorine and its compounds some- times find it helpful to think in terms of acid-base reac- tions in which the fluoride ion (F¯) is donated and ассеpted. (a) Would the acid in this system be the fluoride donor or fluoride acceptor? (b) Identify the acid and base in each of these reactions: CIF;O2 + BF; CIF,O, · BF, -- TiF, + 2 KF – K2[TiF,]arrow_forwardHypochlorous acid (HCIO) is a weak acid. The conjugate base of this acid is the hypochlorite ion (CIO). Wrtie a balanced equation showing the reaction of HClO with water. Include phase symbols. balanced equation: C₁0(aq) +H₂0(1) → HO Write a balanced equation showing the reaction of CIO with water. Include phase symbols. balanced equation:arrow_forward3. Write the products of the reaction between the acid KHP and the base NaOH, and balance the reaction: HKC3H4O4(s) + NaOH(aq) –arrow_forward
- A 0.709 g sample of H2C2O4·2H2O is dissolved in water and titrated to a phenolphthalein endpoint with 27.98 mL of a sodium hydroxide solution. What is the molarity of the NaOH?arrow_forwardIdentify the driving force for the chemical reaction: Pb(NO3)2(ag) + 2N2OH (ag) → Pb(OH)2(s) + 2NaNO3 3(aq) O Formation of a precipitate. O Formation of a water neutralization of an acid and base reduction and oxidation. O There is no driving force.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY