
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please answer the questions thoroughly! Thanks
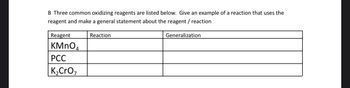
Transcribed Image Text:B Three common oxidizing reagents are listed below. Give an example of a reaction that uses the
reagent and make a general statement about the reagent / reaction
Reagent
KMnO4
PCC
K₂CrO₂
Reaction
Generalization
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can you help me answer this questions please!arrow_forwardThis assignment is designed to help you learn how to determine total costs by combining direct and indirect costs for a particular department. In this GHA, you will be determining the total costs for three departments: (1) Physical Therapy (2) Occupational Therapy and (3) Speech Therapy. To determine the total costs, you will need to add the direct costs to the indirect costs using the template provided below. Please use the example provided in the assignment description if you find that you need some support understanding the required calculations. DIRECT COSTS The direct costs for each department are easily identifiable, because they are the costs that are directly attributable to the department. The direct costs are as follows: Physical Therapy (PT) Occupational Therapy (OT) Speech Therapy (ST) Total Direct Costs INDIRECT COSTS The indirect costs will need to be allocated to Physical Therapy (PT), Occupational Therapy (OT), and Speech Therapy (ST). The indirect costs in this…arrow_forwardWhat are oil in water emulsions ? Give examples from daily lifearrow_forward
- sure your internet connection is working well. Interrupted service can be used as a reason for a retake, only have one attempt. There are no retakes. ot click the "Back" button on your web browser it will kick you out of the exam. e: Make sure to answer all questions. If you close the exam early with answering all questions in your exam. wers that you answered will count as your completed exam and all questions not answered will be marked Question 1 Which of the following best explains why soap and water remove grease? O Soap molecules are large polymers that absorb grease O Soap molecules are ionic and attracted to grease Soap molecules are nonpolar and dissolve in grease O Soap molecules have ionic "heads" that dissolve in water and nonpolar "tails" that dissolve in water O Soap molecules have ionic "heads" that dissolve in grease and nonpolar "tails" that dissolve in water. Quiz saved at 6:52p ch O Oarrow_forwardThis is not a graded question. Subparts 4-5arrow_forward15:32 Sun May 2 Done < AA A session.masteringchemistry.comarrow_forward
- _____ H3PO4 + _____ KOH ----> _____ K3PO4 + _____ H2Oarrow_forwardUnknown Mixture #1 Unknown Mixture #2 Sample caffeine ibuprofen aspirin acetaminoph en Distance to solvent front 14,70 cm 14,70cm 14.70cm1u.70 cm 1나,구 cm |14.구0cm 5. 70 cm 11.90 cm I.85cm Distance Spot(s) traveled 175 cm 13.75cm 11.9 5 cm 6.4 0 cm 13,75 cm 13.75 cm 38 78 1908 .8095 Calculated R:(s) 119 ,9354 812 9 43 S .9354 9354 #1:Aspirin an d lbuprofen Components of Unknowns #2: Ace taminophen, aspirin an d ibu profen D. Additional Exercises 1. Which of the substances tested is most polar? What parts of the structure of this substance is polar? The Sul 2. Which of the substance tested is most non-polar? What parts of the structure of this substance in non-polar?arrow_forwardA 1% solution of hydrochloric acid is required for the procedure. A 5% solution is available. How much of the 5% solution will be needed to make 500 mL of a 1% solution?arrow_forward
- 16- 23arrow_forwardCan someone help with this multistep sytheisis and explain why they got their answers.arrow_forwardCarbon dioxide concentration (parts per million) Examine the graph and answer the following questions. Part A Atmospheric Carbon Dioxide What was the average rate of increase in carbon doxide concentration between 1900 and 19407 410 400 Express you answer in parts per million per year to two significant figures. 300 380 a ? 370 360 ppm/year 350 340 Submit Bequest Anawer 330 320 310 Part 8 Complete previous parts) 300 290 - 1860 1880 1900 1920 1940 1960 1980 2000 2020 Provide Feedback Next> Yeararrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY