
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
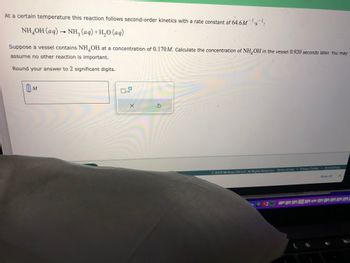
Transcribed Image Text:At a certain temperature this reaction follows second-order kinetics with a rate constant of 64.6M¹s¹:
NH,OH(aq) → NH, (aq)+H,O(aq)
Suppose a vessel contains NH OH at a concentration of 0.170M. Calculate the concentration of NH OH in the vessel 0.920 seconds later. You may
assume no other reaction is important.
Round your answer to 2 significant digits.
0 M
x10
X
S
2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center 1 Accessibility
Show All
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At a certain temperature this reaction follows second-order kinetics with a rate constant of 37.3M¯¹·s¯¹; 2N₂O, (g) → 2N₂O4 (8) + O₂(g) - 5 2 Suppose a vessel contains N₂O5 at a concentration of 0.250M. Calculate the concentration of N₂ in the vessel 0.340 seconds later. You may assume no other reaction is important. 5 Round your answer to 2 significant digits. Marrow_forwardThe initial concentration of reactant in a first-order reaction is 7.6 M. The rate constant for the reaction is 0.27 s−1. What is the concentration of reactant after 5.51 s? Express your answer in M using at least two significant figures. Do not use scientific notation.arrow_forwardAt a certain temperature this reaction follows first-order kinetics with a rate constant of 0.00266 s': H,CO, (aq)→ H,0 (aq) +CO, (aq) Suppose a vessel contains H, CO, at a concentration of 0.740 M. Calculate how long it takes for the concentration of H,CO, to decrease by 89.0%. You may 3. assume no other reaction is important. Round your answer to 2 significant digits.arrow_forward
- At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.0927 s ': 2N,0, (g) – 2N,0, (g) +0, (g) Suppose a vessel contains N,O, at a concentration of 1.02M. Calculate the concentration of N,0, in the vessel 7.10 seconds later. You may assume no other 2 reaction is important. Round your answer to 2 significant digits.arrow_forwardAt a certain temperature this reaction follows second-order kinetics with a rate constant of 0.980 Ms!: 2 HI (g) → H2 (g) + I2 (g) Suppose a vessel contains HI at a concentration of 0.880 M. Calculate the concentration of HI in the vessel 5.30 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits.arrow_forward= 5 = 6 = 7 = 8 9 = 10 = 11 12 13 = 14 At a certain temperature this reaction follows second-order kinetics with a rate constant of 0.0162 M.s: NH,OH (ag) → NH, (aq) +H,0 (aq) Suppose a vessel contains NH,OH at a concentration of 0.700 M. Calculate the concentration of NH,OH in the vessel 590. seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. OM Continue Submit Assignment O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use I Privacy Accessibilityarrow_forward
- At a certain temperature this reaction follows second-order kinetics with a rate constant of 4.91 M s: 2HI (g) - H, (g) +I, (3) Suppose a vessel contains HI at a concentration of 1.33 M. Calculate the concentration of HI in the vessel 2.40 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. ?arrow_forwardAt a certain temperature this reaction follows first-order kinetics with a rate constant of 0.0654 s 2NH₂(g) →1 → N₂(g) + 3H₂(g) Suppose a vessel contains NH3 at a concentration of 0.680M. Calculate the concentration of NH3 in the vessel 9.20 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. M 0.0 X Sarrow_forwardThe rate at which a certain drug is eliminated by the body follows first-order kinetics, with a half life of 75 minutes. Suppose in a particular patient the concentration of this drug in the bloodstream immediately after injection is 1.2 ug/mL. What will the concentration be 150 minutes later? Round your answer to 2 significant digits. ug x10 mLarrow_forward
- The rate at which a certain drug is eliminated by the body follows first-order kinetics, with a half life of 41 minutes. Suppose in a particular patient the concentration of this drug in the bloodstream immediately after injection is 0.35 µg/mL. What will the concentration be 123 minutes later? Round your answer to 2 significant digits. e ml. 0.8arrow_forwardAt a certain temperature this reaction follows first-order kinetics with a rate constant of 0.0667 s¯¹: NH,OH (aq) → NH, (aq)+H,O (aq) Suppose a vessel contains NH4OH at a concentration of 0.360M. Calculate the concentration of NH4OH in the vessel 15.0 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. M x10 X ?arrow_forwardAt a certain temperature this reaction follows second-order kinetics with a rate constant of 0.459 M 1.2N205 (g) - 2N, 04 (8) + 02 (8)Suppose a vessel contains N,O, at a concentration of 0.130M. Calculate the concentration of N, O, in the vessel 95.0 seconds later. You may assume no otherreaction is important.Round your answer to 2 significant digitsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY