
Physics for Scientists and Engineers, Technology Update (No access codes included)
9th Edition
ISBN: 9781305116399
Author: Raymond A. Serway, John W. Jewett
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please solve for (x) using the correct formula and explanation.(Handwritten sol required)
This is the mechanics of materials problem
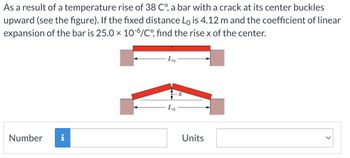
Transcribed Image Text:As a result of a temperature rise of 38 C°, a bar with a crack at its center buckles
upward (see the figure). If the fixed distance Lò is 4.12 m and the coefficient of linear
expansion of the bar is 25.0 × 10-6/Co°, find the rise x of the center.
Lo
Number
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Similar questions
- An aluminum rod 0.500 m in length and with a cross sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 3(H) K. (a) If one-halt of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool, (b) If the circular surface of the upper end of the rod is maintained at 300 K. what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 YV/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forwardA company advertises that it delivers helium at a gauge pressure of 1.72107 Pa in a cylinder of volume 43.8 L. How many balloons can be inflated to a volume of 4.00 L with that amount of helium? Assume the pressure inside the balloons is 1.72105 Pa and the temperature in the cylinder and the balloons is 25.0 .arrow_forwardAssuming the human body is primarily made of water, estimate the number of molecules in it. (Note that water has a molecular mass of 18 g/mol and there are roughly 1024 atoms in a mole)arrow_forward
- Show that =3a , by calculating the infinitesimal change in volume dT of a cube with sides of length L when the temperature changes by dT .arrow_forwardUsing a numerical integration method such as Simpson's rule, find the fraction of molecules in a sample of oxygen gas at a temperature of 250 K that have speeds between 100 m/s and 150 m/s. The molar mass of oxygen (O2) is 32.0 g/mol. A precision to two significant digits is enough.arrow_forwardA mole of gas has isobaric expansion coefficient dV/dT=R/p and isochoric pressure-temperature coefficient dp/dT=p/T . Find the equation of state of the gas.arrow_forward
- The height of the Washington Monument is measured to be 170.00 m on a day when the temperature is 35.0 . What will its height be on a day when the temperature falls to -10.0 ? Although the monument is made of limestone, assume that its coefficient of thermal expansion is the same as that of marble. Give your answer to five significant figures.arrow_forwardWhat is the ratio of the average translational kinetic energy of a nitrogen molecule at a temperature of 300 K to the gravitational potential energy of a nitrogen-molecule-Earth system at the ceiling of a 3-m-tall room with respect to the same system with the molecule at the floor?arrow_forwardSuppose a gas-filled incandescent light bulb is manufactured so that the gas inside the bulb is at atmospheric pressure when the bulb has a temperature of 20.0 . (a) Find the gauge pressure inside such a bulb when it is hot, assuming its average temperature is 60.0 (an approximation) and neglecting any change in volume due to thermal expansion or gas leaks. (b) The actual final pressure for the light bulb will be less than calculated in part (a) because the glass bulb will expand. Is this effect significant?arrow_forward
- As the very first rudiment of climatology, estimate the temperature of Earth. Assume it is a perfect sphere and its temperature is uniform. Ignore the greenhouse effect. Thermal radiation from the Sun has an intensity (the "solar constant" S) of about 1370 W/m2 at the radius of Earth's orbit. (a) Assuming the Sun's rays are parallel, what area must S be multiplied by to get the total radiation intercepted by Earth? It will be easiest to answer in tens of Earth's radius, R. (b) Assume that Earth reflects about 30% of the solar energy it intercepts. In other words, Earth has an albedo with a value of A=0.3 . In terms of S, and R, what is the rate at which Earth absorbs energy from the Sun? (c) Find the temperature at which Earth radiates energy at the same rate. Assume that at the infrared wavelengths where it radiates, the emissivity e is 1. Does your result show that the greenhouse effect is important? (d) How does your answer depend on the area of Earth?arrow_forward(a) Find the density in SI units of air at a pressure of 1.00 atm and a temperature of 20 , assuming that air is 78% N2, 21% O2, and 1% Ar, (b) Find the density of the atmosphere on Venus, assuming that it's 96% CO2 and 4% N2, with a temperature of 737 K and a pressure of 92.0 atm.arrow_forwardFind the ratio f(vp)/f(vrms) for hydrogen gas ( M=2.02 g/mol) at a temperature of 77.0 K.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning


University Physics Volume 1
Physics
ISBN:9781938168277
Author:William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:OpenStax - Rice University

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning