
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
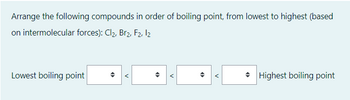
Transcribed Image Text:Arrange the following compounds in order of boiling point, from lowest to highest (based
on intermolecular forces): Cl₂, Br2, F2, 12
Lowest boiling point
<
O
<
Highest boiling point
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (Q43) The normal boiling point of acetone is 56.1°C and its AHvap at its boiling point is 29.1 kJ/mol. What is the vapor pressure (in atm) at 32.7 °C?arrow_forwardPlease help me complete this questionarrow_forwardExplain why the zinc ion should not interfere with absorbance measurements of the copper ion.arrow_forward
- Rank the following compounds in order of decreasing boiling point. ннннн IIIII Н-С-С-С-С-С-Н ннннн O III>I>IV>I| © II>IV>I>III O III>I>II>IV IV>II>I>III © I>III>II>IV нннн н-с-с-с-с-о-н .... нннн || н НIН нн С н Н-С-С-С-С-Н H H H H H нн Ш Η Η Η Η н-с-с-с-C-N-H ..... НЕНН Н IVarrow_forwardWhich of the following compounds has the highest boiling point? OIV ΟΙ III o ll NH2 H IVarrow_forward7. Rank the following compounds in order of increasing boiling point. Ranking 11 OH III HO IV OHarrow_forward
- Compare the boiling points of the various isomeric hydrocarbons shown in the table below. Notice the relationship between boiling point and structure; branched-chain hydrocarbons have lower boiling points than the unbranched isomer. Speculate on possible reasons for this trend. Why might the intermolecular forces be slightly different in these compounds? Compound Boiling Point (°C) 68.9 Hexane 3-Methylpentane 2-Methylpentane 2,3- Dimethylbutane 2,2- Dimethylbutane 63.2 60.3 58.0 49.7 The more branched structure has smaller exposed surface and hence weaker dispersion intermolecular forces. The more branched structure has larger exposed surface and hence weaker dispersion intermolecular forces. The more branched structure has larger exposed surface and hence stronger dispersion intermolecular forces. The more branched structure has smaller exposed surface and hence stronger dispersion intermolecular forces.arrow_forwardRank the following compounds in order of increasing boiling point (lowest to highest) and explain your ranking: OHarrow_forwardRank the following in terms of increasing boiling point. (use 4 for the lowest boiling point and 1 for the highest boiling point) он O. он ноarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY