
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
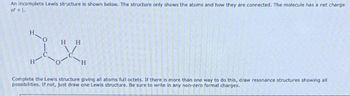
Transcribed Image Text:An incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge
of +1.
HH
XX
H
H
Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all
possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- An incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of zero. H Н—N—С- -о—с—Н H H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges. Click and drag to start drawing a structure.arrow_forwardPlease draw the Lewis structure of the polyatomic ion PO2- without forgetting the resonance structure. Please break down the steps clearly with details so that I can follow along. Thank you for your help!arrow_forwardWrite the Lewis structure for SO, where the central sulfur atom obeys the octet rule. Do not include any charges on your drawing. Show only one drawing (do not include any additional resonance structures). H olN F Br CI Morearrow_forward
- What is wrong with the Lewis structure depicted below? [Image description: A Lewis structure with a O atom singly bound to three H atoms and a C atom. The C atom also has two electron pairs and another single bond to a H atom.] H н—о—с — н H Multiple answers: Multiple answers are accepted for this question Select one or more answers and submit. For keyboard navigation... SHOW MORE V a At least one atom has less than an octet b Separation of formal charge persists C Formal charges are not minimized d Row 2 element exceeds octet e Incorrect central atomarrow_forwardCalculate the formal charge for the (fictional) elements below using the information given below, for the polyatomic ion shown below the table. Complete the sentence with your answers. Element Symbol Number of Valence Electrons Electronegativity Value A 1 0.53 X 1 0.62 E 2 0.91 R 3 1.32 M 4 1.62 T 5 2.70 D 6 3.05 L 7 3.21arrow_forwardConsider the Lewis structure of L2Z shown below where neither L nor Z are capable of forming an expanded octet. Without changing the central atom, how many total resonance structures, including the one shown below, are possible? Group of answer choices 4 3 2 1arrow_forward
- Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure |N—N=N] : 0: :0-C H .. I :Ö: H 1 O-H + Is the proposed Lewis structure reasonable? Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are: Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are: * Yes. No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are: * * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "0,0".arrow_forwardWrite a Lewis structure for each of the following polyatomic ions. Show all bonding valenceelectron pairs as lines and all nonbonding valence electron pairs as dots. For those ions thatexhibit resonance, draw the various possible resonance forms.a. phosphate ion, PO4 3-b. chlorate ion, ClO3-arrow_forwardI. For each of the following reactions, the elements combine through transfer of one or more electrons, resulting in a completely filled outermost shell form each atom. Draw Lewis structures for each atom, assigning the correct number of valence electrons and the correct changes. 1) Ва + 21 2) 2K + O 3) ЗАГ + Br » Ва 12 → K2 O > Al Br3arrow_forward
- There are four exceptions to the octet rule. State them below: A hydrogen atom will never carry an octet. Instead, a hydrogen atom carries a _____ which means it is in possession of _____ electrons. Boron (B) and Beryllium (Be) can exist with _______ (fewer or more) than an octet of electrons. Non-metals from period 3 and above can exist with _______ (fewer or more) than an octet of electrons. Free radicals are molecules that can have ______________ electrons.arrow_forwardDraw Lewis structure(s) for the carbonate lon (CO₂). If there are equivalent resonance structures, draw all of them. n D co₂2: 0 . Draw one structure per sketcher box, and separate added sketcher boxes with the symbol. Do not include overall lon charges or formal charges in your drawing. Do not draw double bonds to oxygen unless they are needed in order for the central atom to obey the octet rule. ● 6 # H Ⓒ CH, CHO: 0 Y Chemic b Draw Lewis structure(s) for the acetaldehyde molecule (CH₂CHO). If there are equivalent resonance structures, draw all of the POLICE 81 MEDITE HARA (4) Y Draw one structure per sketcher box, and separate added sketcher boxes with the symbol. Do not include overall ion charges or formal charges in your drawing. Do not draw double bonds to oxygen unless they are needed in order for the central atom to obey the octet rule. ARQQA 000-ZIF www HEADING Chartlkoodn MES DE A V Ja remove 000-n [ MacBook Airarrow_forwardA valid Lewis structure of O carbon tetrafluoride phosphate ion O silicon tetrafluoride O nitrogen trifluoride selenium tetrafluoride cannot be drawn without violating the octet rule.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY