Question
T14e please help me with my reviewer, I can’t get the value for the step 2, also kindly give me a detail solution so that I can’t I understand it thank you
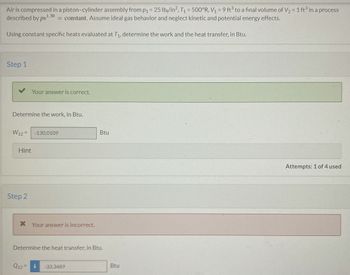
Transcribed Image Text:Air is compressed in a piston-cylinder assembly from p₁ = 25 lb/in², T₁ = 500°R, V₁ = 9 ft³ to a final volume of V₂ = 1 ft³ in a process
described by pv1.30 = constant. Assume ideal gas behavior and neglect kinetic and potential energy effects.
Using constant specific heats evaluated at T₁, determine the work and the heat transfer, in Btu.
Step 1
Determine the work, in Btu.
Your answer is correct.
W12= -130.0109
Hint
Step 2
* Your answer is incorrect.
Q12=
Determine the heat transfer, in Btu.
Btu
-33.3489
Btu
Attempts: 1 of 4 used
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Figure y (cm) 50 B Integration path 50 x (cm) 1 of 1 Part A What is the line integral of B between points i and f in (Figure 1)? Assume that B = 0.10 T. Express your answer using two significant figures. Ψ—| ΑΣΦ i B. ds = Submit Provide Feedback Request Answer ? T narrow_forwardS Can 6 PAR к Торс K Unit K In x K Moti = Cop K Unit S Spee S Topo S Math Micr eb.kamihq.com/web/viewer.html?source-filepicker&document_identifier=137VZR5BZOVSAIMOA55WU555_CvJ9NacO + 100 P e Interpreting Graphs Answer the questions following the graphs on each side Dietance va. Time 2 7 10 11 12 13 14 Time in soconds 1. From 1 second to 2 seconds, how fast is the object traveling. (Take the difference in distance and divide it by the time in between the 2 distances) 2. Is the object going as fast between 9 and 12 seconds as it is between 1 and 4 seconds? How can you tell? 3. What is the motion of the object between 4 and 6 seconds? acerarrow_forwardSolve the following problem step by step using the given formulas (show steps clearly )arrow_forward
- Please solve fassst.arrow_forwardIf we have stabdard deviations for x and y data and we have mean values for x and y, how can we evaluate the standard deviation of (1-x/y).m ? (m is a constant value).arrow_forwardNeed help on finding the values of R_k , R_0 and S . If you can show me how to solve it step by step , thanks in advance .arrow_forward
- t Eg=15N and Eo=l", Find Fymn The Smallest Possible Vahek Of minj ty in N Find Fxmar The largest possible Value or Fx, in N maxarrow_forwardSolve the following problem (NOTE: Use any reasonable DATA not given in the problem.)....arrow_forwardCan you show me how to best analyze this graph to solve for the problem?arrow_forward
arrow_back_ios
arrow_forward_ios