
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
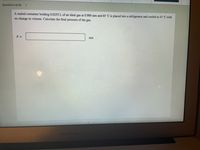
Transcribed Image Text:Question 4 of 28
<>
A sealed container holding 0.0255 L of an ideal gas at 0.989 atm and 65 °C is placed into a refrigerator and cooled to 43 °C with
no change in volume. Calculate the final pressure of the gas.
P =
atm
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A 9.00 L tank at 2.1 °C is filled with 7.71 g of boron trifluoride gas and 9.11 g of carbon dioxide gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: boron trifluoride partial pressure: atm mole fraction: carbon dioxide partial pressure: atm Total pressure in tank: atmarrow_forwardA 7.00 L tank at -7.0 °C is filled with 2.65 g of chlorine pentafluoride gas and 7.83 g of boron trifluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to 3 significant digits. gas mole fraction ? olo chlorine pentafluoride Ar boron trifluoridearrow_forwardA 10.0 L tank at 18.9 °C is filled with 13.1 g of sulfur hexafluoride gas and 6.12 g of carbon monoxide gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: sulfur hexafluoride partial pressure: atm mole fraction: carbon monoxide partial pressure: I atm atm Total pressure in tank:arrow_forward
- A 6.00 L tank at -2.4 °C is filled with 2.01 g of sulfur tetrafluoride gas and 6.11 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to 3 significant digits. gas sulfur tetrafluoride sulfur hexafluoride mole fraction 0 0 X Ś 0 000 13 Ararrow_forwardA 5.00 L tank at 7.96 °C is filled with 10.8 g of chlorine pentafluoride gas and 4.88 g of dinitrogen difluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. ol. mole fraction: x10 chlorine pentafluoride Ar ? partial pressure: atm mole fraction: dinitrogen difluoride partial pressure: atm Total pressure in tank: atmarrow_forwardA 6.00 L tank at -6.3 °C is filled with 12.2 g of carbon monoxide gas and 10.5 g of chlorine pentafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to 3 significant digits. gas mole fraction ? olo carbon monoxide 18 Ar chlorine pentafluoridearrow_forward
- A 9.00 L tank at 3.79 °C is filled with 8.94 g of sulfur tetrafluoride gas and 13.3 g of sulfur hexafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. sulfur tetrafluoride sulfur hexafluoride mole fraction: partial pressure: mole fraction: partial pressure: Total pressure in tank: 0 0 0 atm atm atm Xarrow_forwardA 9.00 L tank at 6.02 °C is filled with 8.81 g of sulfur hexafluoride gas and 10.5 g of dinitrogen difluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: sulfur hexafluoride partial pressure: atm mole fraction: dinitrogen difluoride partial pressure: atm Total pressure in tank: atmarrow_forwardA 6.00 L tank at 19.2 °C is filled with 3.42 g of boron trifluoride gas and 6.16 g of carbon dioxide gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: x10 boron trifluoride partial pressure: atm mole fraction: carbon dioxide partial pressure: atm Total pressure in tank: || atmarrow_forward
- A 9.00 L tank at 8.6 °C is filled with 11.0 g of sulfur tetrafluoride gas and 9.86 g of dinitrogen difluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Round each of your answers to 3 significant digits. gas sulfur tetrafluoride dinitrogen difluoride mole fraction 1 0 X Sarrow_forwardA 6.00 L tank at 7.53 °C is filled with 5.91 g of sulfur hexafluoride gas and 7.97 g of boron trifluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas, and the total pressure in the tank. Round each of your answers to 3 significant digits. mole fraction: sulfur hexafluoride partial pressure: ? atm mole fraction: boron trifluoride partial pressure: atm Total pressure in tank: atmarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY