
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
A paper plant is proposed for a location 1 km upwind from a town. It will emit 40 g/s of hydrogen sulfide, which has an odor threshold of about 0.1 mg/m3. Assume that winds at 10 m above ground are 2.5 m/s, and at the effective stack height they typically vary from 4 – 10 m/s blowing toward the town. If it is summer day with a few broken clouds, using the provided equation,
- What stack height should be used to assure concentrations are no more than 0.1 times the odor threshold at the near edge of town (1 km away)? To be conservative, the stack will be designed assuming no plume rise.
- Will any buildings (e.g., the country cabin or the doghouse on the far edge of town) experience higher concentrations than a residence at the 1 km boundary? Briefly explain your answer.
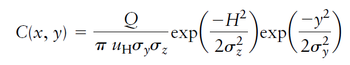
Transcribed Image Text:C(x, y)
=
п ино Uz
ехр
-Н2
на
20²2²
exp
-22
20²
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps with 16 images

Knowledge Booster
Similar questions
- A 10 cm thick piece of beef steak will be frozen in the freezer room -40 ° C. This product has a moisture content of 73%, a density of 970 kg / m³, and a thermal conductivity (frozen) of 1.1 W / (m K). Estimate the freezing time. using the Plank equation. This product has an initial freezing temperature of -1.75 ° C, and the movement of air in the freezing room gives a convective heat transfer coefficient of 15 W / (m² K). t f = hourarrow_forwardA piece of beef steak 7 cm thick will be frozen in the freezer room -30 ° C. This product has a moisture content of 73%, a density of 970 kg / m³, and a thermal conductivity (frozen) of 1.1 W / (m K). Estimate the freezing time. using the Plank equation. This product has an initial freezing temperature of -1.75 ° C, and the movement of air in the freezing room gives a convective heat transfer coefficient of 15 W / (m² K). t f = ... hour.arrow_forward1. Consider a 1000 MW power plant located in a rural area with 15 ton/day SO2 emissions from a 100 m high stack. The velocity and temperature of the stack gases lead to an effective stack height of 50 m above the physical stack. Estimate the ground level concentration as a function of distance downwind under the following conditions. The emissions are into a clear daytime atmosphere with wind (at 10 m) of 5 m/s. b. The emissions are into a clear nighttime atmosphere with wind (at 10 m) of 2 m/s. The conditions of a. except there is a strong elevated inversion at an altitude of 200 m. a. С.arrow_forward
- On a day when wind is blowing over a lake (T(water) =20°C) at 5 m/s, which side of the air-water interface will be rate-limiting on the flux of Ethyl benzene (i.e. va or vw)?arrow_forwardIf solar radiation intensity is 700 W/m² in NJ, how much solar energy arrives at a solar collector with an area of 2 m² in 12 hours? If the solar collector can convert 33% of solar energy collected to electricity, how much electricity (kWh) will be generated?arrow_forwardchemical engineering A power plant emits 216 g of SO2 every hour. The wind recorded on the same day is 5 m s-1, and the atmospheric stability class is A for the clear summer afternoon. There is no plume rise, and the stack height is 30 m. The values of σy and σz are 215 m and 450 m, respectively. Determine: the concentration of SO2 0.7 km downwind along the plume centreline at ground level.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The