
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
Transcribed Image Text:A 89% (4) E Wed 12:31 PM
Edit
View History Bookmarks People Tab Window Help
Chrome
File
G standard formation reaction fo x
G convert kj to j- Google Search x
ALEKS - Jacqueline Hoppenrey x
C Search Textbook Solutions | Ch x
A www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQIHQRDYV_6Ux63SypJXz0Coxvwqgg4JkWI7IUnyRjVNsBjUR5oMNI9R1NdBbh5VEqEfXiSbDcv772zARqYw50M1_Zaol-D2.
Jacqueline v
O THERMOCHEMISTRY
Using specific heat capacity to find temperature change
OO C
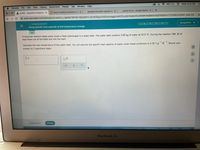
A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 3.60 kg of water at 32.0 °C. During the reaction 106. kJ of
heat flows out of the flask and into the bath.
-1
·K
Round your
Calculate the new temperature of the water bath, You can assume the specific heat capacity of water under these conditions is 4.18 J•g
answer to 3 significant digits.
dlb
Explanation
Check
2021 McGraw-Hill Education. All Rights Reserved Terms of Use Privacy Accessibility
MacBook Air
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A 20.94 g sample of a metal is heated to 99.4°C in a hot water bath until thermal equilibrium is reached. The metal sample is quickly transferred to 100.0 g of water at22.0°C contained in a calorimeter. The thermal equilibrium temperature of the metalsample plus water mixture is 24.6°C. What isthe specific heat of the metal? Express the specific heat with the correct number of significant figures.arrow_forwardThe specific heat of a certain type of metal is 0.128 J/(g.°C). What is the final temperature if 305 J of heat is added to 63.2 g of this metal, initially at 20.0 °C? °C Tfinal ||arrow_forwardAn insulated container is used to hold 42.7 g of water at 20.5 Celsius. A sample of copper weighing 9.60 g is placed in a dry test tube and heated for 30 minutes in a boiling water bath at 100.0 Celsius. The heated test tube is carefully removed from the water bath with laboratory tongs and inclined so that the copper slides into the water in the insulated container. Given that the specific heat of solid copper is 0.385 J/(g.Celsius), calculate the maximum temperature of the water in the insulated container after the copper metal is added.arrow_forward
- An 18.89 g sample of neon gas in a container with a movable wall is initially at a temperature of 320 K and a pressure of 1.0 atm (which matches the external pressure of 1.0 atm). The container is then heated to 405 K, and its volume increases until its pressure is once again at 1.0 atm. How much work was done on the system in this process? Report your final answer in units of J.arrow_forwardLearning Goal: To understand the concepts of heat capacity, specific heat, and molar heat capacity. Heat capacity, C, is the amount of energy required to raise the temperature of a substance by exactly one degree Celsius. The energy needed to warm an object increases as the mass of that object increases. We see this in our everyday life. For example, we know that it takes much more energy to heat a large tank of water than a small cup. Because of this dependence on mass, experimentally determined heat capacities are always reported in terms of the amount of the substance that is heated. One method is to report how much energy it takes to raise the temperature of one mole of a substance by exactly one degree Celsius. This value is the molar heat capacity, which has the symbol Cp. The molar heat capacity is given in the units J mol- °C-1.A second method is to report how much energy it takes to raise the temperature of one gram of a substance by exactly one degree Celsius. This value is…arrow_forwardA chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 9.10 kg of water at 25.1 °C. During the reaction 59.0 kJ of heat flows out of the bath and into the flask. - 1 Calculate the new temperature of the water bath. You can assume the specific heat capacity of water under these conditions is 4.18 J.g Round your answer to 3 significant digits. °C x10 ?arrow_forward
- The temperature of an object increases by 43.5 °C when it absorbs 3829 J of heat. Calculate the heat capacity of the object. C = The mass of the object is 367 g. Use the table of specific heat values to identify the composition of the object. Substance Specific heat (J/(g. °C)) gold 0.129. silver 0.240 iron 0.444 aluminum 0.900 The object is composed of O aluminum. silver. gold. iron. J/°Carrow_forwardA chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 5.30 kg of water at 26.9 °C. During the reaction 78.1 kJ of heat flows out of the flask and into the bath. -1 -1 Round your Calculate the new temperature of the water bath. You can assume the specific heat capacity of water under these conditions is 4.18 J∙g ¹·K answer to 3 significant digits. 1°C x10 X Ś ?arrow_forwardA group of students place 15.41g of a metal into a test tube. They place the test tube into a beaker of boiling water until the temperature of the metal was 89.8°C. Then they quickly place the heated metal into a beaker of 10.47g of water with an initial temperature of 16.4°C. The temperature of the mixturecame to rest at 26.1°C. What is the specific heat of the metal used? (CH2O = 4.18 J/g°C)arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY