
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
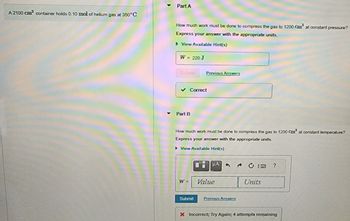
Transcribed Image Text:A 2100 cm³ container holds 0.10 mol of helium gas at 350°C.
Part A
How much work must be done to compress the gas to 1200 Cm² at constant pressure?
Express your answer with the appropriate units.
▸ View Available Hint(s)
W = 220 J
✓ Correct
Part B
Previous Answers
How much work must be done to compress the gas to 1200 Cm³ at constant temperature?
Express your answer with the appropriate units.
► View Available Hint(s)
W =
Submit
μA
Value
Previous Answers
Units
?
X Incorrect; Try Again; 4 attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A cylinder contains 0.250 mol of carbon dioxide (CO₂) gas at a temperature of 27.0°C. The cylinder is provided with a frictionless piston, which maintains a constant pressure of 1.00 atm on the gas. The gas is heated until its temperature increases to 127.0°C. Assume that the CO₂ may be treated as an ideal gas. Part A How much work is done by the gas in this process? Express your answer in joules. — ΑΣΦ W = Submit Part B On what is this work done? The work is done on the piston. The work is done on the cylinder. Submit Part C Request Answ AU = What is the change in internal energy of the gas? Express your answer in joules. ΑΣΦ Submit Request Answer Request Answer P Pearson W ? ? J Jarrow_forwardFor a planet to have an atmosphere, gravity must be sufficient to keep the gas from escaping The escape speed a particle needs to escape the earth's gravitational attraction is 1.1 x 10 m/s. The motion of projectiles never depends on mass, so this escape speed applies equally to rockets and to molecules in the earth's upper atmosphere. Part A At what temperature does the rms speed of nitrogen molecules equal the escape speed? Express your answer in kelvins. ΜΕ ΑΣΦΑ T- Submit Part B T- Request An At what temperature does the mms speed of hydrogen molecules equal the escape speed? Express your answer in kelvins. VAZO Submit ? K Karrow_forwardI Rev Part A How much work is done by the gas in the process shown in the figure? (Figure 1) Express your answer in joules. V AZO ? W = J Submit Request Answer Figure 1 of 1 > Provide Feedback Р (КРа) 400- 200 i V (cm³) 100 200 300arrow_forward
- In an engine, an almost ideal gas is compressed adiabatically to half its volume. In doing so, 2710 J of work is done on the gas. Part A ▼ How much heat flows into or out of the gas? Express your answer with the appropriate units. ? Q = Value Units Submit Request Answer Part B What is the change in internal energy of the gas? Express your answer with the appropriate units. HẢ ? AU = Value Units Submit Request Answerarrow_forwardAn ideal gas at 20°C consists of 2.2 x 1022 atoms. 3.2 J of thermal energy are removed from the gas. Part A What is the new temperature in °C? Express your answer in degrees Celsius. DA ΑΣφ T = °C Submit Request Answerarrow_forwardPart C Some hydrogen gas is enclosed within a chamber being held at 200°C with a volume of 0.0250 m³. The chamber is fitted with a movable piston. Initially, the pressure in the gas is 1.50 × 106 Pa (14.8 atm). The piston is slowly extracted until the pressure in the gas falls to 0.950 x 106 Pa. What is the final volume V₂ of the container? Assume that no gas escapes and that the temperature remains at 200°C. Enter your answer numerically in cubic meters. ► View Available Hint(s) V₂ = Submit [5] ΑΣΦ ? m³arrow_forward
- A diver observes a bubble of air rising from the bottom of a lake (where the absolute pressure is 3.50 atm) to the surface (where the pressure is 1.00 atm). The temperature at the bottom is 4.0°C, and the temperature at the surface is 23.0°C. Part A What is the ratio of the volume of the bubble as it reaches the surface to its volume at the bottom? Enter your answer numerically. VE ΑΣΦ V2 V₁ Submit Part B Request Answer Would it be safe for the diver to hold his breath while ascending from the bottom of the lake to the surface? Submit No. This increase in volume of air in the lungs would be dangerous. Yes. This increase in volume of air in the lungs is safe enough for human body. Provide Feedback ? Request Answerarrow_forwardI need solutions question Barrow_forwardA cylinder contains 0.250 mol of carbon dioxide (CO₂) gas at a temperature of 27.0°C. The cylinder is provided with a frictionless piston, which maintains a constant pressure of 1.00 atm on the gas. The gas is heated until its temperature increases to 127.0°C. Assume that the CO₂ may be treated as an ideal gas. Express your answer in joules. AU = Submit Part D Submit How much heat was supplied to the gas? Express your answer in joules. Part E W₁ = ΑΣΦ | Submit Request Answer ΑΣΦ Request Answer Request Answer < Return to Assignment Provide Feedback www How much work would have been done if the pressure had been 0.50 atm? Express your answer in joules. VE ΑΣΦ P Pearson = ? ? J ? J Jarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON