
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
None
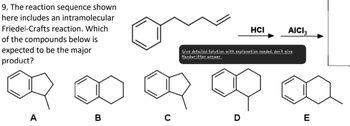
Transcribed Image Text:9. The reaction sequence shown
here includes an intramolecular
Friedel-Crafts reaction. Which
of the compounds below is
expected to be the major
product?
HCI
AICI 3
Give detailed Solution with explanation needed, don't give
Handwritten answer
A
B
C
D
E
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Similar questions
- Did I do this correctly?arrow_forwardwhat are the coefficients missing on the following equation?_KN03+_H2CO3--->_K2CO3+_HNO3 O (2,1,1,2) O (0,0,0,2) (2,2,0,0) O (0,0,0,0) O (2,2,2,0)arrow_forwardName these organic compounds: Η H H H | | | Η· C C C C C=H | | | | | H H H H H — H Η structure — — H H - C C 1 1 Η - Η - - Η - H | H — C - Η | H namearrow_forward
- Research studies have found that the price elasticity of demand for bread is equal to -0.40 and the price elasticity of demand of butter is equal to -1.13. Holding everything else constant, if the price of bread and the price of butter each increase by 10 percent: (a) Will the revenue from sales of bread increase or decrease? (b) Will the revenue from sales of butter increase or decrease?arrow_forwardUse and modify some/all the reactions provided to form the indicated overall reaction according to Hess' Law. • If a reaction is not used in your Hess' Law process, include that indication by multiplying the reaction by "0" • If no modification is needed and the reaction is applied as written below, include that indication by multiplying the reaction by "1" • Use whole numbers and simple fractions (such as "2" or "1/2", not 0.5) and negative signs (such as "-2") where needed Then calculate and report the enthalpy change (in kJ) for the overall reaction in the indicated blank, including units with your written value and rounded to 1 decimal place. Overall Reaction: CS2(1) + 302(g) → CO2(g) + 2SO2(g) Rxn # Reaction ΔΗ° (kJ) 1 C(s) → C(g) 716.67 2 C(s) + O2(g) → CO2(g) -393.5 3 3Fe(s) +202(g) → Fe3O4(s) -1118 4 C(s) + 2S(s) → CS2(1) 87.9 5 CH4(g) + O2(g) → CO₂(g) + H₂O(g) -882.0 6 S(s) + O2(g) → SO2(g) -296.8 N2(g) + 4H2(g) + Cl2(g) → 2NH4Cl(g) -629.1 7arrow_forwardWith the information provided answer Carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY