
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chemical Equilibrium:
Write the K formula for the following reactions.
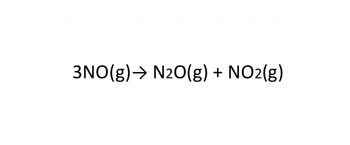
Transcribed Image Text:3NO(g) → N2O(g) + NO2(g)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
Hi! Is this correct?
Kc = [N2O] [NO2] / [NO]^3
Solution
by Bartleby Expert
Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
Hi! Is this correct?
Kc = [N2O] [NO2] / [NO]^3
Solution
by Bartleby Expert
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the following equilibrium system: 2SO2 + O2 --> 2SO3 Into a 2.00 Liter container, 0.500 mol of SO3, was placed and heated to 300°C. After equilibrium was established, the container was found to have 0.300 moles of SO3 present along with the other gases. Calculate the equilibrium concentration of each molecule and the equilibrium constant for this reaction at this temperature.arrow_forwardSulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a 125. L tank with 11. mol of sulfur dioxide gas and 35. mol of oxygen gas, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 7.7 mol. Calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to 2 significant digits. K. = |arrow_forwardThe molecule borneal can rearrange to a different form called isoborneal in a process called isomerization. A container initially contains only borneal gas at a pressure of 553 torr. At equilibrium, the pressure of borneall is 500.0 torr and the pressure of isoborneol is 53.0 torr. Is the isomerization reaction exergonic or endergonic? Calculate the value of the equilibrium constant for the reaction, borneal ⇌ isoborneol.arrow_forward
- Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor, and in the liquid form it is easily transported. An industrial chemist studying this reaction fills a 1.5 L flask with 1.7 atm of ammonia gas and 2.1 atm of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of water vapor to be 0.77 atm. Calculate the pressure equilibrium constant for the combustion of ammonia at the final temperature of the mixture. Round your answer to 2 significant digits. K = ☐ P x10arrow_forwardCalculating an equilibrium constant from a partial equilibrium compositionarrow_forwardO KINETICS AND EQUILIBRIUM Calculating equilibrium composition from an equilibrium constant Suppose a 500. mL flask is filled with 1.9 mol of NO,, 0.10 mol of N0 and 1.7 mol of CO,. The following reaction becomes possible: 2' NO,(g)+CO(g) -NO(g)+CO,(g The equilibrium constant K for this reaction is 4.70 at the temperature of the flask. Calculate the equilibrium molarity of NO. Round your answer to two decimal places. M TRADIS Explanation Check 2020 McGraw-Hill Education. All Rights Reserved. Terms of Use | Privacy | Acces NOV 242 PAGES étv 4 MacBook Air IIarrow_forward
- What does it mean for a chemical reaction to reach equilibrium? The rates of the forward and reverse reactions are equal. The concentrations of the reactants and products are also equal. The rates of the forward and reverse reactions are increasing. The concentrations of the reactants and products are equal. The rates of the forward and reverse reactions are equal. The concentrations of the reactants and products are constant. The rates of the forward and reverse reactions are increasing. The concentrations of the reactants and products are also increasing.arrow_forwardWrite balanced net ionic equations for the following reversible reactions. Then write the corresponding equilibrium constant, k, for each. 1) Hydrogen gas reacts with nitrogen has to form ammonia gas and steam. 2) Nitrogen gas reacts with solid sodium carbonate and solid carbon to produce carbon monoxide gas and solid sodium cyanide (NaCN).arrow_forwardCalculating an equilibrium constant from a partial equilibrium composition Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor, and in the liquid form it is easily transported. An industrial chemist studying this reaction fills a 2.0 L flask with 1.2 atm of ammonia gas and 3.7 atm of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of water vapor to be 1.3 atm. Calculate the pressure equilibrium constant for the combustion of ammonia at the final temperature of the mixture. Round your answer to 2 significant digits. K = [] P x10 X ? olo 18 Ar 8.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY