1A. Identify the species getting oxidized or reduced, as well as the oxidizing and reducing agents in the following reactions. Mn2+ + BiO3 → MnO4- + Bi3+ Oxidized: __________________ Oxidizing agent: __________________ Reducing agent: __________________ Reduced: __________________ 1B. The following are isomers of each other. I. pentane II. 2,2,-dimethylpropane III. 2-methylbutane IV. 2,3-dimethylhexane A. Alkanes are nonpolar. B. Alkanes are non-flammable. C. Every carbon in an alkane has four bonds. D. Alkanes contain only C and H atoms. E. Alkanes do not contain carbon-carbon double or triple bonds. 1C. What is the correct name for (picture is attatched) A. 3-ethyl-2-methylbutane B. 3-ethyl-2-methylpentane C. 2-ethyl-1,3-dimethylbutane D. 1,2,3-trimethylpropane E. 4,5-dimethyl-2-hexane
1A. Identify the species getting oxidized or reduced, as well as the oxidizing and reducing agents in the following reactions.
Mn2+ + BiO3 → MnO4- + Bi3+
Oxidized: __________________
Oxidizing agent: __________________
Reducing agent: __________________
Reduced: __________________
1B.
The following are isomers of each other.
I. pentane
II. 2,2,-dimethylpropane
III. 2-methylbutane
IV. 2,3-dimethylhexane
A.
B. Alkanes are non-flammable.
C. Every carbon in an alkane has four bonds.
D. Alkanes contain only C and H atoms.
E. Alkanes do not contain carbon-carbon double or triple bonds.
1C.
What is the correct name for (picture is attatched)
A. 3-ethyl-2-methylbutane
B. 3-ethyl-2-methylpentane
C. 2-ethyl-1,3-dimethylbutane
D. 1,2,3-trimethylpropane
E. 4,5-dimethyl-2-hexane

Oxidized species will release electrons.
Reduced species will gain electrons.
Oxidizing species will oxidized another substance therefore itself gets reduced.
Reducing species will reduced another substance therefore itself gets oxidized.
1A :
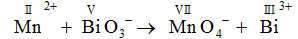
Mn here releases electron and goes from +2 to +7 oxidation state. So ,
BiO3- will accept electron and goes from +5 to +3 oxidation state.
Oxidized : MnO4-
Oxidizing agent: BiO3-
Reducing agent: Mn(II)
Reduced: Bi3+
Step by step
Solved in 4 steps with 2 images









