
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
There are several possible Lewis structures for the molecule N2O as shown below.
See attachment
For Structure A what are the formal charges on atoms:
1 , 2 , and 3 ?
For Structure B what are the formal charges on atoms:
1 , 2 , and 3 ?
For Structure C what are the formal charges on atoms:
1 , 2 , and 3 ?
Compare the formal charges that you calculated for each structure.
What is the letter of the BEST Lewis structure for N2O?
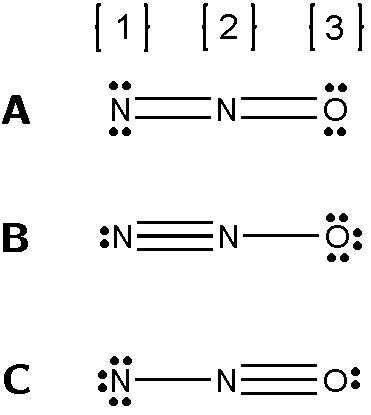
Transcribed Image Text:[1} [2} [3}
EN=Ö
A
B
EN-ö:
:N
:N-NEo:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the BEST Lewis structure for hydrogen cyanide, HCN, the formal charge charge on H is __________, the formal charge on C is___________ , and the formal charge on N is _________. Indicate formal charge with a sign followed by a numerical charge (e.g. -2, +1) unless the charge is 0, then enter 0.arrow_forwardThe formal charge is the "charge" an element would have in a molecule or ion if all of the bonding electrons were shared equally between atoms. Use the References to access important values if needed for this question. We can draw three inequivalent Lewis structures for dinitrogen monoxide, N₂O . The concepts of formal charge and electronegativity can help us choose the structure that is the most significant representation. 1. Assign formal charges to the elements in each of the structures below. Formal Charge N₁ N₂ :N,=N,-O: || A N=N,=Ô N-N,=O: C 2. The structure that contributes most significantly to the overall electronic structure of N₂O is ↑ Formal Chargearrow_forwardConsider the SO 3 molecule. How many valence electrons does the molecule have? How many lone pairs of electrons are on the central atom? How many lone pairs of electrons in total are on the substituent atoms? How many resonance structures can be drawn for the molecule? If the ion doesn't exhibit resonance, indicate "1" as only one structure can be drawn for the molecule.arrow_forward
- a. Write a Lewis structure that obeys the octet rule for the following species. Assign the formal charge for the central atom of XeO4. If multiple resonance structures exist, use one that does not involve an expanded valence. Formal charge: b. Write a Lewis structure that obeys the octet rule for the following species. Assign the formal charge for the central atom of clO3¯. If multiple resonance structures exist, use one that does not involve an expanded valence. Formal charge:arrow_forwardDraw a Lewis structure for NH4+. Based on this Lewis structure, the calculated value for the formal charge on the nitrogen atom is O 1+ 0 0 O 1- O 4- O 4+arrow_forward12arrow_forward
- Draw three resonance structures for N2O (N is central) that follow the octet rule and assign formal charges. Rank the structures from best to worst and draw the hybrid form of the best two structures.arrow_forwardOne of the Lewis structures that can be drawn for the N₂O molecule is: :N=N-O: What are the formal charges on the N (end), N (center), and O atoms in this structure? 0, -1, +1 0, +1, -1 O 0, 0, 0 +1, +1, -2 +1, 0, -1arrow_forwardThe value of Ka1 of hypophosphorous acid, H3PO2, is nearly the same as the Ka1 of phosphoric acid, H3PO4. Draw a Lewis structure for phosphoric acid that is consistent with this behavior. Draw the Lewis structure with the formal charges minimized. Do not include formal charges in your structure. Include hydrogen atoms and nonbonding electrons in the structure.arrow_forward
- Write resonance forms that describe the distribution of electrons in each of these molecules or ions. a) nitric acid, HNO3 (N is bonded to an OH group and two O atoms) b) benzene, C6H6: c)the formate ion:arrow_forwardGive handwritten answerarrow_forwardFor a molecule of XeCl3H a) Draw the Lewis structure then predict the shape of the compound and draw the shape of the compound b) Determine if the molecule is polar or nonpolar and explain how you made that determination. If there is a dipole moment, draw it.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY