
Concept explainers
Interpretation:
The series of equations for the preparation of each of the given compound from the designated starting material using suitable organic or inorganic reagents is to be written.
Concept introduction:
The conjugate base of alkyne acts as a good nucleophile. On reaction with primary
In hydration of alkynes, a molecule of water gets added to a molecule of alkyne.Hydrogen atom gets attached to the less substituted triple bonded carbon atom, and hydroxyl groupgetsbonded to the more substituted triple bonded carbon atom and forms a
Geminal and vicinal dihalides, on reaction with a strong base, undergo double dehydrohalogenation and form alkynes.
On halogenation, alkynes form vicinal dihalides and, on reaction with hydrogen halides, they form geminal dihalides in two steps.
Primary alcohol can be converted to primary alkyl bromide by reaction with sodium bromide in acidic condition.
Answer to Problem 36P
Solution:
a) 
b) 
c) 
d) 
e) 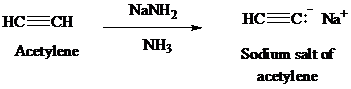

f) 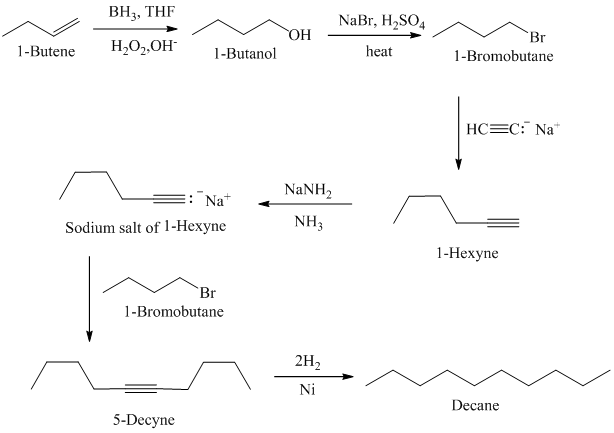
g) 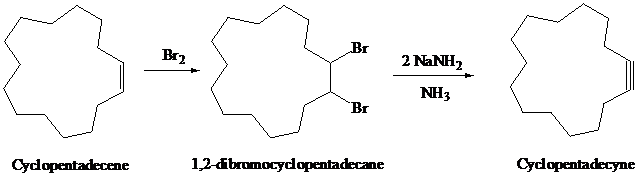
h) 
Explanation of Solution
a)
The
The synthesis of

b)
The
The synthesis of

c)
The
The synthesis of

d)
Acetylene on treatment with sodium amide, gives sodium salt of acetylene. The sodium salt of acetylene on treatment with ethyl bromide gives
The synthesis of

e)
Acetylene on treatment with sodium amide, gives sodium salt of acetylene. In presence of organoboron,
The synthesis of
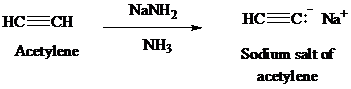

f)
In presence of organoboron,
The synthesis of

g)
Cyclopentadecene on halogenation with
The synthesis of
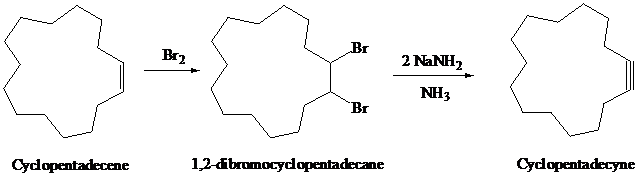
h) 
In presence of base sodium amide, the given alkyne gives sodium salt of alkyne. The sodium salt of alkyne on treatment with bromomethane adds one methyl group to alkyne, which on partial hydrogenation with Lindlar catalyst gives required cis alkene.
The synthetic route is as follows:

Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry - Standalone book
- 1. The substance to be analyzed – is white, highly soluble in water salt. Flame coloration test gives weakly-violet color. Interaction with concentrated sulfuric acid leads to evolving of reddish-brown gas with pungent odour. Mixing of salt water solution with chlorine water in presence of trichloromethane changes of color of organic layer to orange. What kind of substance is this? Explain you answer, provide chemical equationsarrow_forwardAn organic compound S reacted with ozone to give compounds Q and R. Both Q and R react with NaOH/I2 to produce a yellow precipitate with antiseptic smell. Q reacts with HCN to produce the compound CH3C(CH3)OHCN while R gives a silver mirror with Tollens reagent. I) write the structural formulae and names of Q,R,S and the Yellow Precipitate. II) write equations to show the reactions Q with NaOH/I2 and R with H^+/Cr2O7^2-arrow_forwardOnly one of the chlorine atoms in the molecule, 3,4-dichloronitrobenzene, will undergo nucleophilic substitution. Indicate which position will react and provide the expected product for the given reaction using reaction intermediates (resonance structures).arrow_forward
- Suggest a method of preparing ethyl benzene, starting with benzene and ethylene as the only organic reagents.arrow_forwardDraw a structure for each of the following compounds. You may use any acceptable style of drawing (skeletal, condensed, structural, etc.) Part 1) isopropyl chloride Part 2) cis-1-sec-butyl-3-fluorocycloheptane Part 3) (R)-8-bromo-5-iodo-1,3-octadiynearrow_forward2. Using structural diagrams to explain the synthesis of butyl ethanoate from an alkane and an alcohol. Make sure you have the names of the substances and the reaction conditions of the reactions of the steps that consists your synthesis HC-C H- styrene HH ethane-1,2-diol OH НО. JOL -0-H benzene-1.4-dicarboxylic acid.arrow_forward
- For the reaction shown, in blank '1', use IUPAC nomenclature and name the major product of the reaction shown. In blank '2; use IUPAC nomencature and name the minor product of the reaction shown. In the 3rd blank, select the name of the type of reaction. Is it halogenation, hydration, hydrogenation, or hydrohalogenation. HBr ?arrow_forwardComplete the following sequence of reactions including the products of each step. Please be clear with your writing. etanol = ethanolarrow_forwardWrite the products formed as a result of the reactions given below.arrow_forward
- Provide a chemical equation including all reagent(s) and condition(s) to synthesis the following compounds as major product from suitable alkene. a) 2,3-dibromo-2-methylpentane b) 2-chloro-1,1-dimethylcyclohexanearrow_forwardWhen 30 cm3 of a gaseous hydrocarbon was exploded with 210cm3 of oxygen the residual gases occupied volume of 150cm3. After shaking the residual gases with excess aqueous sodium hydroxide, the final volume was 30cm3.(All volumes were measured at room temperature and pressure.) The hydrocarbon has a number of isomers. Draw the displayed formulae and name 2 of the isomers of the hydrocarbon that illustrate each of the following types of isomerism below (i.e. 2 isomers must be drawn for each type of isomerism) (i) chain (ii)functional grouparrow_forwardDraw structural formulas for organic products A and B in the window below. CI Li CH,OH CH3CHCH3 pentane • Draw only products having the organic portion of the original alkyl halide. • Draw carbon-lithium bonds using the single bond tool. If a structure has a copper-lithium bond, do not draw the lithium. • Separate products from different steps using the → sign from the drop-down menu. opy astearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





