
Concept explainers
Interpretation:
The retrosynthesis analysis and the actual synthesis are to be written for the preparation of each target molecule from the given starting molecule.
Concept introduction:
>The retrosynthesis is a reaction that involves
on acid catalyzed dehydration, an alcohol gives the corresponding more substituted alkene as the major product.
>The alkene, on reaction with halogen in water, undergoes addition of halogen and hydroxyl group across the double bond in a way that the halogen atom is bonded to the less substituted double bonded carbon atom and hydroxyl group is bonded to the more substituted double bonded carbon atom.
>Alkenes react with hydrogen halide and yield haloalkane.
>Alkenes, on hydration, form more substituted alcohol.
>Alkenes, on hydroboration-oxidation, form less substituted alcohol.
>Alkenes, in presence of peroxy acid, undergo
In presence of a strong base,
Answer to Problem 50P
Solution:
The retrosynthetic analysis and the actual synthesis for the preparation of each target molecule from the given starting compound are as follows:
a)
Retrosynthesis:
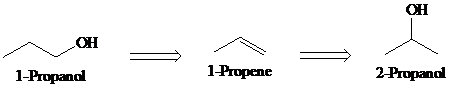
Synthesis:
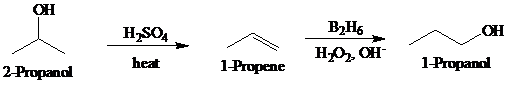
b)
Retrosynthesis:
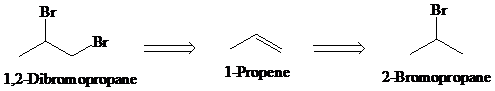
Synthesis:
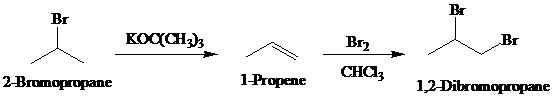
c)
Retrosynthesis:
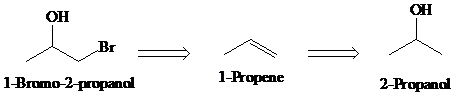
Synthesis:
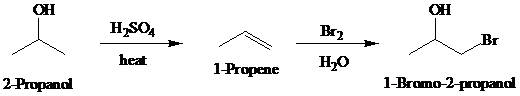
d)
Retrosynthesis:
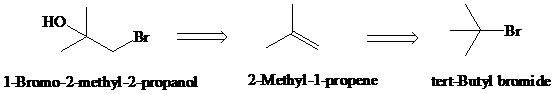
Synthesis:
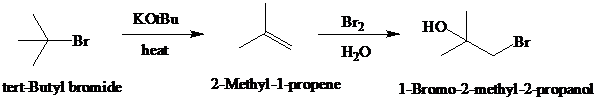
e)
Retrosynthesis:
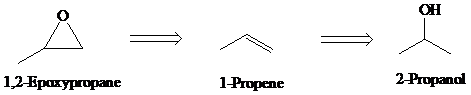
Synthesis:

f)
Retrosynthesis:
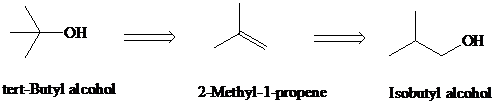
Synthesis:

g)
Retrosynthesis:
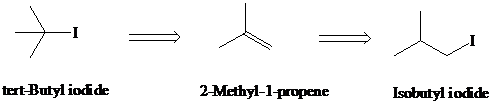
Synthesis:

h)
Retrosynthesis:
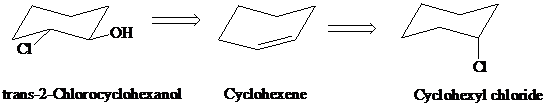
Synthesis:

Explanation of Solution
a)
Retrosynthetic analysis for the target molecule
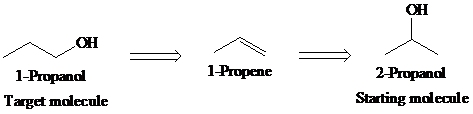
The actual synthetic route for the above retrosynthesis is as follows:
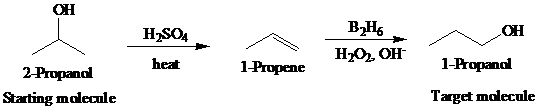
b)
Retrosynthetic analysis for the target molecule
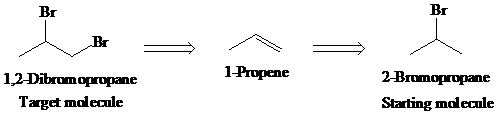
The actual synthetic route for the above retrosynthesis is as follows:
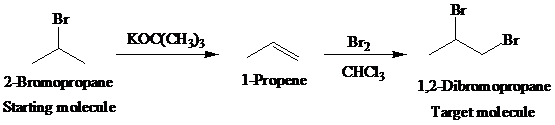
c)
Retrosynthetic analysis for the target molecule
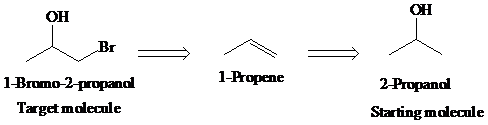
The actual synthetic route for the above retrosynthesis is as follows:
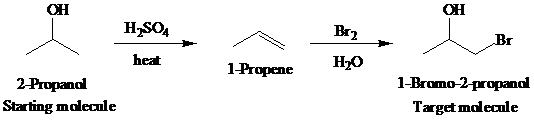
d)
Retrosynthetic analysis for the target molecule
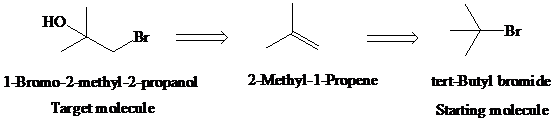
The actual synthetic route for the above retrosynthesis is as follows:
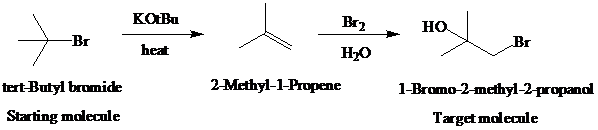
e)
Retrosynthetic analysis for the target molecule
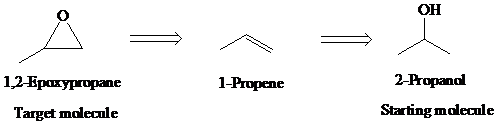
The actual synthetic route for the above retrosynthesis is as follows:
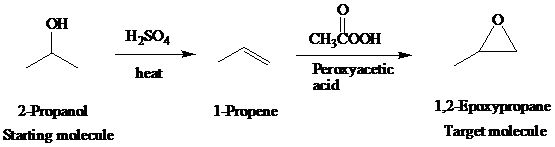
f)
Retrosynthetic analysis for the target molecule
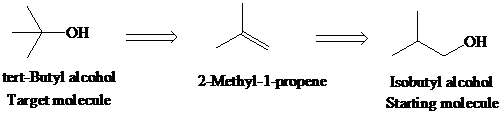
The actual synthetic route for the above retrosynthesis is as follows:
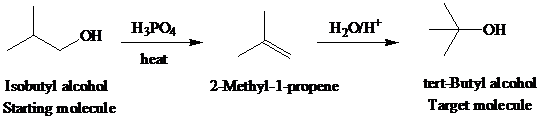
g)
Retrosynthetic analysis for the target molecule
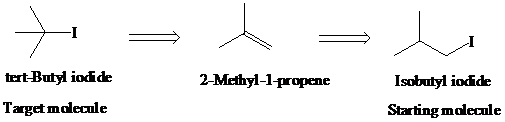
The actual synthetic route for the above retrosynthesis is as follows:
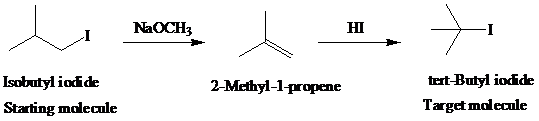
h)
Retrosynthetic analysis for the target molecule

The actual synthetic route for the above retrosynthesis is as follows:

Therefore, the retrosynthesis and synthesis reactions for the given compounds were proposed.
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry - Standalone book
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardAldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forwardWrite an equation for the reaction (if any) of benzaldehyde with each of the following reagents: - a) Tollen's reagent b) hydroxylamine c) ethyl magnesium bromide, then H3O+ d) phenylhydrazine e) HCN f) excess methanol, H+ g) LiAlH4 h) ethylene glycol, H+arrow_forward
- 4) Complete the following reaction by filling in the necessary reagents. OH 1) 2) OH OHarrow_forwardIdentify the best reagents to complete the following reaction. Options are included.arrow_forwardReaction of 1-butanol with periodane gives Butanal Propanoic acid 2-Butanone Butanoic acid as the major product.arrow_forward
- Acetals are formed from the reaction of two alcohols with a carbonyl under acidic conditions. Acetal formation is faster with 1,2-ethanediol than with two methanol molecules. Choose the factor that explains the difference in reaction rates. A) The reaction with 1,2-ethanediol has a lower AH (enthalpy) of reaction. B) The reaction with 1,2-ethanediol has a higher AH (enthalpy) of reaction. C) The reaction with 1,2-ethanediol has a more favorable entropy of reaction.arrow_forwardWhat will be the major organic product from the dehydration of 2-propanol in the presence of a strong acid and high temperature? A) 2-propanone B) 1-propene C) propane D) propanal E) propane-1,2-diolarrow_forwardWhat reactions and reagents can be used to make phenol from benzene if electrophilic aromatic substitution reactions are excluded and benzene is the only source of carbon?arrow_forward
- Order the compound according to increasing boiling point: a) 3-pentanone b)2-methylcyclopentene c) 3-hydroxybutanoic acid d)methoxy-2-butane e)2-ethyl-1-butanol f)N-ethyl-2-pentanaminearrow_forward15.) Which of the following esters, upon hydrolysis, produces a diol as one of the products? A) hydroxymethyl hexanoate D) cyclohexyl hexanoate B) 2-methylpropyl butanoate E) diethyl decanedioate C) ethyl 3-iodopentanoate F) cyclopropyl benzoatearrow_forwardStarting from cyclohexanol, show how to prepare: Show all reagents and molecules synthesized along the way after each step. You do not have to use curved arrows.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
