
Concept explainers
(a)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(a)
Explanation of Solution
The given organic compound is as follows:
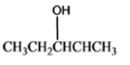
The name of the compound is 2-butanol.
Here, hydroxyl group is attached to the secondary carbon thus, it is a secondary alcohol.
(b)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(b)
Explanation of Solution
The given organic compound is as follows:
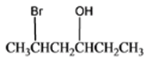
The name of the above compound is 5-bromo-3-hexanol.
Here, the hydroxyl group is at secondary carbon thus, it is a secondary alcohol.
(c)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(c)
Explanation of Solution
The given organic compound is as follows:
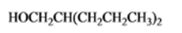
The name of the compound is 2-propyl-1-pentanol.
Now, there is hydroxyl group at primary position thus, it is a primary alcohol.
(d)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(d)
Explanation of Solution
The given organic compound is as follows:
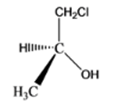
The name of the compound is (S)-1-chloro-2-propanol. There is a hydroxyl group at secondary carbon thus, it is a secondary alcohol.
(e)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(e)
Explanation of Solution
The given organic compound is as follows:
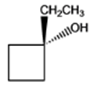
The name of the above compound is 1-ethylcyclobutanol. The hydroxyl group is at tertiary carbon thus, it is a tertiary alcohol.
(f)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(f)
Explanation of Solution
The given organic compound is as follows:
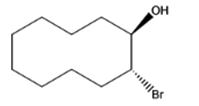
The name of the compound is (1R,2R)-2bromocyclodecanol.
In the above compound, the hydroxyl group is at secondary position thus, this is a secondary alcohol.
(g)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(g)
Explanation of Solution
The given organic compound is as follows:
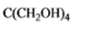
The name of the compound is 2, 2-bis(hydroxylmethyl)propane-1,3-
(h)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(h)
Explanation of Solution
The given organic compound is as follows:
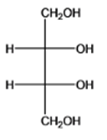
The name of the molecule is (2S,3R)-2,3-dihydroxybutane-1,2-diol. The hydroxyl group at first and fourth carbon atoms is primary and the other two hydroxyl groups are at second and third carbon atoms which are secondary.
(i)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(i)
Explanation of Solution
The given organic compound is as follows:
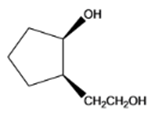
The name of the compound is cis-2-(2-hydroxylethyl)cyclopentanol. Here, the hydroxyl group is at secondary carbon atom of the cyclopentyl ring and the other hydroxyl group is attached to the primary carbon atom.
(j)
Interpretation: The name of the given alcohol needs to be determined, the stereochemistry (if present) needs to be indicated and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(j)
Explanation of Solution
The given compound is as follows:
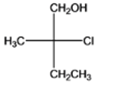
The name of the compound is (2R)-2-chloro-2-methyl-1-butanol. The hydroxyl group is at primary carbon atom thus, it is primary alcohol.
(k)
Interpretation: The name of the given alcohol and its all possible stereoisomers needs to be determined and it needs to be labeled as primary, secondary and tertiary alcohol.
Concept Introduction: An alcohol can be classified as primary, secondary and tertiary deepening on the number of hydrogen atoms attached to the C atom to which the hydroxyl group is attached. If the number of H atoms are two, the alcohol is primary in nature, if there is only 1 H atom attached, then the alcohol is secondary and if there is no H atom attached to the C atom then the alcohol is tertiary.
(k)
Explanation of Solution
The given structure is as follows:
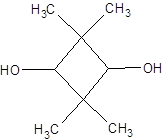
Since, there is no chiral center thus, there will be no possible stereoisomers.
The name of the given alcohol is 2, 2, 4, 4-tetramethylcyclobutane-1, 3-diol. Both the −OH groups are on the C atom with 1 H atom each thus, both are secondary alcohols.
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- Draw a structural formula for the major organic product of the following reaction: CH₂Cl₂ CH3CH=CHCH₂CH(CH3)2 + Br₂arrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardDraw a structural formula for the alkene you would use to prepare the alcohol shown by hydroboration/oxidation. H3C CH3 CH3CHCCH3 ОН You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one.arrow_forward
- Draw a structural formula for the alkene you would use to prepare the alcohol shown by hydroboration/oxidation. OH ļ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. + Sn [F ?arrow_forwardDraw a structural formula for the product formed upon hydroboration/oxidation of the alkene below. CH3 • Use wedge and hash bonds ONLY for rings. • Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer.arrow_forwardWhich structural formulas represent cis isomers of 1,2-dimethylcyclohexane? H H H II IV I and II III and IV I and IV Il and IIIarrow_forward
- Draw the structural formula for 3-chlorohexanenitrile. O O O O O CT CH 3 CH ₂ CH CH ₂ CH ₂ CN CH 3 CH ₂ CH ₂ CH CH ₂ CH ₂ CN CI CH3 CH₂ CH₂ CH2 CH CH ₂ CN CI CH3 CH2 CH₂ CH CH ₂ CN दा CH3 CH ₂ CH CH ₂ CH ₂ CH ₂ CNarrow_forward4-Chlorobut-1-ene is the name of which among the following alkenes? O CH2CI-CH2=CH-CH2 O CH2CI-CH2-CH-CH2 O CH2CI=CH2-CH=CH2 O CH2CI-CH2-CH=CH2arrow_forwardCH,-C-CHCH, Classify each compound as an aldehyde or a ketone. Give the common name for each ketone. CH,CH,CH,-C-H CH,CH,CH,-C-CH,CH,CH, CH, CH;-C-CHCH, Name the following organic compound containing oxygen. CH, O CH, 3, | CH,CH-C-CHCH, || CH,CH,CH,-C-CH,CH, CH3 || CH,CH,CH,CH-Ĉ-H CH3 CH,CH,CH-C–Harrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





