
Concept explainers
(a)
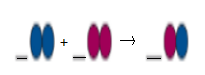
Interpretation: To balance the following reaction by adding coefficients and identify the type of reaction.
Concept Introduction:
Mass can neither be created nor be destroyed.
A
Types of chemical reaction:
- Combination reaction: Here two or more reactants combines to form a single product
- Decomposition product: In this type of reaction, one reactant is breakdown into two or more products
- Displacement reaction: This is a type of reaction where more reactive element displaces the less reactive element.
- Double-displacement reaction: In this type of reaction, two compounds react in such a way that cations and anions of two reactants interchange their place.
Balanced equation will be:
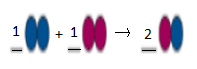
This is a type of combination reaction.
In the given reaction, the reactant contains two blue atoms and two pink atoms.
So,
Product should contain 2 blue atoms and 2 pink atoms.
But in product side, we have a molecule having only one blue atom and 1 pink atom. Therefore, we need to multiply the product with 2 to make it balance.
In this chemical reaction,
2 reactants are combined to form a single product. Hence, it is a type of combination reaction.
(a)
Answer to Problem 7.101UTC
Balanced equation will be:
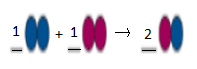
This is a type of combination reaction.
Explanation of Solution
In the given reaction, the reactant contains two blue atoms and two pink atoms.
So,
Product should contain 2 blue atoms and 2 pink atoms.
But in product side, we have a molecule having only one blue atom and 1 pink atom. Therefore, we need to multiply the product with 2 to make it balance.
In this chemical reaction,
2 reactants are combined to form a single product. Hence, it is a type of combination reaction.
(b)
Interpretation: To balance the following reaction by adding coefficients and identify the type of reaction.
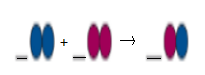
Concept Introduction:
Mass can neither be created nor be destroyed.
A chemical reaction should be well balance i.e. the number of atoms of each element in reactant side must be equal to number of atoms in product side.
Types of chemical reaction:
- Combination reaction: Here two or more reactants combines to form a single product
- Decomposition product: In this type of reaction, one reactant is breakdown into two or more products
- Displacement reaction: This is a type of reaction where more reactive element displaces the less reactive element.
- Double-displacement reaction: In this type of reaction, two compounds react in such a way that cations and anions of two reactants interchange their place.
(b)
Answer to Problem 7.101UTC
Balanced equation will be:
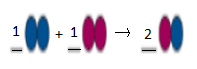
This is a type of decomposition reaction.
Explanation of Solution
In the given reaction, the reactant side contains 2 blue atoms and 1 pink atom.
So, product side should contain 2 blue atoms and 1 pink atom.
But in product side, we have two different products as:
First product has 2 blue atoms and second molecule has 2 pink atoms.
If we multiply the reactant and first product by 2, then reaction will be balance as now both reactants and product have 4 blue atoms and 2 pink atoms respectively.
Here one reactant breaks down into two products. Thus, it is a type of decomposition reaction.
Want to see more full solutions like this?
Chapter 7 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





