
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 5.2, Problem 5.2E
Interpretation Introduction
Interpretation: Molecular orbitals in figure 5.3(a) should be labeled as g or u.
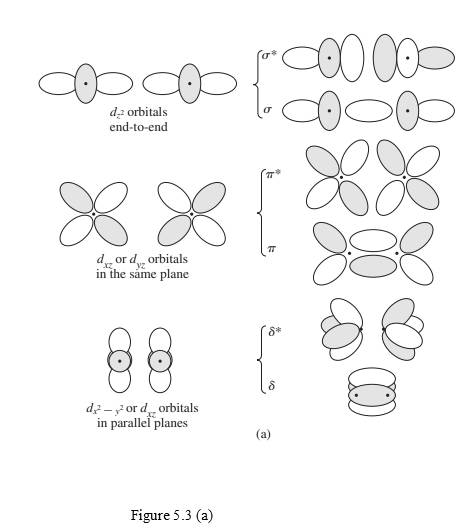
Concept Introduction:
“gerade” (g) used to named orbitals which are symmetric to inversion.
“ungerade” (u) used to named orbitals which are anti-symmetric to inversion.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a bond-line structure that best matches the given 3D representation?
3) Combine any Atomic orbitals to form Molecular orbitals assuming they fulfill the phase and energy requirement.
Construct MO diagram (include AOs, MOs, pictures of each, electrons, HOMO, LUMO, B.O., and comparative energies, etc.). REMOVE an ELECTRON to form CATION
Please only do part 3. The first image shows the problem
5.
(2 points) Consider ethylene, and acetylene
ethylene
acetylene
(a) Draw three-dimensional diagrams of each molecule. Label the hybridization of each
carbon, and show all sigma and pi bonds.
(b) Which of these molecules has the shortest bond length? Which has the longest? In
each of these molecules, where is the largest electron density located in space?
Chapter 5 Solutions
Inorganic Chemistry
Ch. 5.1 - Repeat the process in the preceding example for...Ch. 5.2 - Prob. 5.2ECh. 5.3 - Use a similar approach to the discussion of HF to...Ch. 5.4 - Sketch the energy levels and the molecular...Ch. 5.4 - Using the D2h character table shown, verify that...Ch. 5.4 - Using orbital potential energies, show that group...Ch. 5.4 - Prob. 5.7ECh. 5.4 - Prob. 5.8ECh. 5.4 - Prob. 5.9ECh. 5.4 - Use the projection operator method to derive...
Ch. 5.4 - Determine the types of hybrid orbitals that are...Ch. 5.4 - Determine the reducible representation for all the...Ch. 5 - Expand the list of orbitais considered in Figures...Ch. 5 - On the basis of molecular orbitals, predict the...Ch. 5 - On the basis of molecular orbitals, predict the...Ch. 5 - Compare the bonding in O22,O2 and O2 Include Lewis...Ch. 5 - Although the peroxide ion, O22 and the acetylide...Ch. 5 - High-resolution photoelectron spectroscopy has...Ch. 5 - a. Prepare a molecular orbital energy-level...Ch. 5 - a. Prepare a molecular orbital energy-level...Ch. 5 - NF is a known molecule a. Construct a molecular...Ch. 5 - The hypofluorite ion, OF can be observed only with...Ch. 5 - Prob. 5.11PCh. 5 - Although KrF+ and XeF+ have been studied, KrBr+...Ch. 5 - Prepare a molecular orbital energy level diagram...Ch. 5 - Methylene, CH2 plays an important role in many...Ch. 5 - Beryllium hydride, BeH2 is linear in the gas...Ch. 5 - In the gas phase, BeF2 forms linear monomeric...Ch. 5 - For the compound XeF2 do the following: a. Sketch...Ch. 5 - TaH5 has been predicted to have C4v symmetry, with...Ch. 5 - Describe the bonding in ozone, o3 on the basis of...Ch. 5 - Describe the bonding in SO3 by using group theory...Ch. 5 - The ion H3+ has been observed, but its structure...Ch. 5 - Use molecular orbital arguments to explain the...Ch. 5 - Prob. 5.23PCh. 5 - Prob. 5.24PCh. 5 - The isomenc ions NSO (thiazate) and SNO...Ch. 5 - Apply the projection operator method to derive the...Ch. 5 - Apply the projection operator method to derive the...Ch. 5 - A set of four group orbitals derived from four 3s...Ch. 5 - The projection operator method has applications...Ch. 5 - Although the cl2+ ion has not been isolated, it...Ch. 5 - BF3 is often described as a molecule in which...Ch. 5 - SF4 has C2v symmetry. Predict the possible...Ch. 5 - Consider a square pyramidal AB5 molecule. Using...Ch. 5 - Prob. 5.34PCh. 5 - For the molecule PCl5 : a. Using the character...Ch. 5 - Molecular modeling software is typically capable...Ch. 5 - Prob. 5.39PCh. 5 - Calculate and display the orbitals for the linear...Ch. 5 - Prob. 5.41PCh. 5 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Diborane, B2H6 , has the structure shown. a. Using...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. In the molecular orbital (MO) scheme for frontier orbitals of benzene, answer the following: a) how many of the carbons in benzene are sp²/p hybridized? b) how many p-n electrons does this system have? c) how many T,7* MOs can be constructed from the carbon p-orbitals? d) labeling these yn (n= 1 to ....) increasing in energy, how many vertical nodes do the following MOs have ? Y2,43 ¥4,Y5 e) on the hexagonal skeleton to right, draw the molecular orbital for y3, or y4. Are these MOs bonding, non-bonding or antibonding? Clearly show/tell why. f) in benzene, what orbital(s) is/are the HOMO? yn = g) what is the (value of the) aromatic stabilization energy for benzene? h) is benzene more or less stable than 1,3,5-cyclohexatriene? more less i) in two words, what is the structure of benzene? the LUMO? yn = kJ/mol (circle one)arrow_forwardMatch the following cartoons of molecular orbitals with the correct Greek letter label. If it is an antibonding orbital, be sure that the label you choose is consistent with that determination.arrow_forwardA student argues the the two nitrogen’s in the compound below are sp2 hybridized, but atom a is still more basic than atom b. Is the student correct? Explain.arrow_forward
- (a) Which of the AF3 molecules in Fig. 1 will have a nonzero dipole moment? Explain. b) Which of the AF4 molecules in Fig. 2 will have a zero dipole moment? Explain.arrow_forwardCircle all the isolated (not conjugated) pi bond(s).arrow_forwarda) Consider a molecule of acetone cyanohydrin, shown below. What is the hybridization and molecular geometry at CA, CB, and O? (b) For Carbon B (CB) and nitrogen (N), draw orbital energy diagrams showing the orbitals involved in bonding after hybridization. Be sure to fill the orbitals appropriately with valence electrons. c) In your diagrams in part (b), which orbitals are involved in sigma and pi bonding? Orbitals Involved in Sigma Bonding Orbitals Involved in Pi Bonding 2p sp sp2 sp3 2p sp sp2 sp3 (d) In a sample containing many molecules of acetone cyanohydrin (and no other substances), what type(s) of intermolecular forces could be present? Choose all that apply. Covalent Bonds Hydrogen Bonds Dipole-Dipole Forces London Dispersion Forcesarrow_forward
- Label the following molecular orbitals as o or a orbitals and briefly explain. Be sure indicate the subscript (g or u) and superscript (with or without *), but you do not need to specify the number before o or T. a) b) d)arrow_forward*2.16 For each of the species below, circle the most reactive lone pair or bond and indicate the type of frontier orbital that it represents (n, π, or σ). A B с H₂C CH₂ D NEC +OEN H NH2arrow_forwardMolecule C shown below contains a group called an allene. An allene has the structure of one central carbon being doubly bonded to two other carbons (C=C=C). Please draw out all of the p-orbitals that are involved in the pi-system for molecule C. b) The p-orbital on carbon 1 in molecule C are not in conjugation with the p-orbital on carbon 2 (ie the p- orbitals are not pointing in the same direction and there is no overlap). Why does this happen?arrow_forward
- (please give clear handwritten answer) Draw a complete molecular orbital diagram for LiF. Explain that Energy levels of the atomic orbitals and what kind of character the bonding and antibonding orbitals display.arrow_forwardBF3 is a molecule with ["3", "2", "1", "4"] electron domains giving it a ["linear", "trigonal planar", "tetrahedral"] electron domain geometry. It has ["3", "4", "2", "1"] bonded atoms and ["1", "2", "0", "3"] lone pairs. BF3 will exhibit a ["linear", "trigonal planar", "trigonal pyramidal", "tetrahedral", "bent"] molecular geometry.arrow_forwardA)mark any formal charges on the structures below, and provide the hybridization for any atom with an arrow pointed to it. B) below draw the various Newman projections listed if looking down the C2--> C3 bondarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning