
Interpretation:
The suprafacial or antarafacial stereochemistry of the following sigmatropic reaction by order [x, y] is,
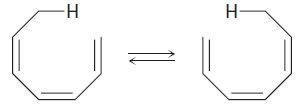
Concept introduction:
A sigmatropic rearrangement is a process in which a sigma-bonded substituent atom or group migrates across a pie electron system from one position to another. So, A sigmatropic reaction in organic chemistry is a pericyclic reaction wherein the net result is one σ-bond is changed to another σ-bond.
Sigmatropic rearrangements are concisely described by an order term [i, j], which is defined as the migration of a σ-bond adjacent to one or more π systems to a new position (i-1) and (j-1) atoms removed from the original location of the σ-bond. [3] When the sum of i and j is an even number, this is an indication of the involvement of a neutral, all C atom chain. An odd number is an indication of the involvement of a charged C atom or of a heteroatom lone pair replacing a C = C double bond. Thus, [1, 5] and [3, 3] shifts become [1, 4] and [2, 3] shifts with heteroatoms, while preserving symmetry considerations.
If the migrating group remains on the same face of the π system, the shift is known as suprafacial, while if the migrating group transfers to the opposite face is called an antarafacial shift.
Trending nowThis is a popular solution!

Chapter 30 Solutions
Organic Chemistry
- The transformation takes place via two sequential pericyclic reactions. Identify the two reactions and give a critical explanation whether the reactions are allowable or not. Explain the stereochemistry.arrow_forwardA tautometric keto (A) – enol(B) equilibrium may be formulated as follows (A) (B) CH3 – COH ß-----------àCH2 – CHOH Given the following bond energies: C – H = 435kJ mol-1 C – C = 368 C==C = 610 C – O = 357 kJmol-1 C==O = 748 kJmol-1 O – H = 462 kJ mol-1 Calculate the enthalpy change in going from Keto form (A) to the enol form (B)arrow_forwardFor each one of the following reactions, show step by step mechanism of the reactions and indicate the stereochemistry of the products and their relationshiparrow_forward
- Predict the products and include stereochemistyarrow_forwardDehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.arrow_forwardAddition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=Carrow_forward
- ORGANIC CHEMISTRY Justify the stereoselectivity of the following Diel Alders intramolecular reaction:arrow_forwardGive the process of Determining the Mechanism and Stereochemistry inNucleophilic Substitution ?arrow_forwardIdentify the following pericyclic reaction; explain the course, product distribution and stereochemistry of the reaction. Where the first product is produced 70% and the second product is produced 30%.arrow_forward
