
Concept explainers
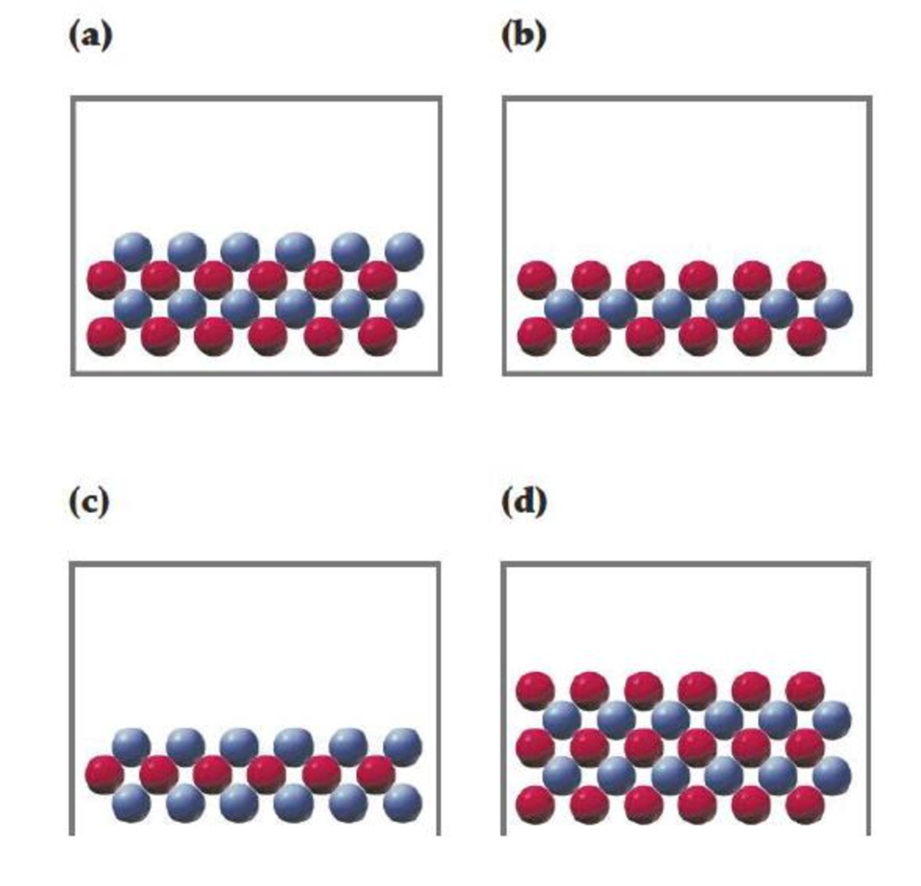
In the following drawings, red spheres represent cations and blue spheres represent anions. Match each of the drawings (a)–(d) with the following ionic compounds:
- (i) Ca3(PO4)2
- (ii) Li2CO3
- (iii) FeCl2
- (iv) MgSO4
Interpretation:
For the given compounds correct representation of diagramhas to be chosen.
Explanation of Solution
To choose: Correct representation of diagram for given compounds.
Given,
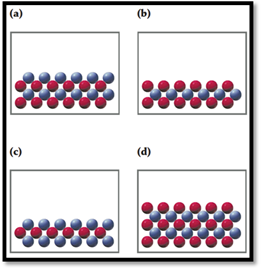
Figure 1
Consider red sphere as cation and blue sphere as anion.
The ratio of cation (
The ratio of cation (
The ratio of cation (
The ratio of cation (
For the given compounds correct representation of diagram was chosen.
Want to see more full solutions like this?
Chapter 3 Solutions
General Chemistry: Atoms First
- 2-98 Explain how the ionization energy of atoms changes when proceeding down a group of the Periodic Table and explain why this change occurs.arrow_forwardThe following ionic compounds are found in common household products. Name each of the compounds:(a) Ca(H2PO4)2(b) FeSO4(c) CaCO3(d) MgO(e) NaNO2(f) KIarrow_forwardName each ionic compound. In each of these compounds, the metal forms more than one type of ion. (a) CrCl2 (b) C:CI3 (c) SnO2 (d) Pbl2arrow_forward
- Write formulas for ionic compounds. (a) What is the formula of the ionic compound expected to form between the elements sodium and fluorine? (b) What is the formula of the compound formed between the ions Co²+ and SO₂²? (c) What ions make up the ionic compound Zn(CN)₂? Cation formula Anion formulaarrow_forwardAnswer true or false. (a) For Group 1A and Group 2A elements, the name of the ion each form is simply the name of the element followed by the word ion; for example, Mg21 is named magnesium ion. (b) H2 is named hydride ion. (c) The nucleus of H1 consists of one proton and one neutron. (d) Many transition and inner transition elements form more than one positively charged ion. (e) In naming metal cations with two different charges, the suffix -ous refers to the ion with a charge of 11 and -ic refers to the ion with a charge of 12. (f) Fe31 may be named either iron (III) ion or ferric ion. (g) The anion derived from a bromine atom is named bromine ion. (h) The anion derived from an oxygen atom is named oxide ion. (i) HCO3 2 is named hydrogen carbonate ion. (j) The prefix bi- in the name “bicarbonate” ion indicates that this ion has a charge of 22. (k) The hydrogen phosphate ion has a charge of 11, and the dihydrogen phosphate ion has a charge of 12. (l) The phosphate ion is PO3 42. (m)…arrow_forwardGive systematic names to the following compounds:(a) KNO2 (b) Sr(MnO4)2(c) MgCr2O7 (d) NaH2PO4(e) BaCl2 (f) NaClO3arrow_forward
- Name the following molecular compounds: (a) NI3arrow_forwardChapter 2 3+ 2- + 2.62 Predict the chemical formulas of the compounds formed by the following pairs of ions: (a) Cr³+and Br, (b) Fe³+ and O², (c) Hg and CO (d) Ca and CIO, (e) NH and PO lon Ca2+ Fe 2+ A1³+ 2.64 Complete the table by filling in the formula for the ionic compound formed by each pair of cations and anions, as shown for the first pair. oz - NO3 SOA 504² 2- 3- Aso ASO4 04 + Na Na 2.66 Which of the following are ionic, and which are molecular? (a) PF (b) Nal, (c) SC, (d) Ca(NO₂) (e) FeCl (f) LaP, (g) COCO (h) ₂0 2.78 The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds: (a) NO, (b) NO, (c) NO₂ (d) N₂O (e) N04arrow_forwardMany chemical names are similar at first glance. Give the for-mulas of the species in each set: (a) ammonium ion and ammo-nia; (b) magnesium sulfide, magnesium sulfite, and magnesiumsulfate; (c) hydrochloric acid, chloric acid, and chlorous acid; (d) cuprous bromide and cupric bromidearrow_forward
- Which of the following sets contains an ionic compound, a molecular compound, and an acid, in that order? (A) Al2O3, B2O3, CH3OH; (B) CaCl2, NH4Cl, HCl; (C) CH3F, COCl2, HOCl; (D) CoCl2, COCl2, HClO2.arrow_forwardIllustrated are four ions — A, B, X, and Y— showing their relativeionic radii. The ions shown in red carry positive charges: a2+ charge for A and a 1+ charge for B. Ions shown in blue carrynegative charges: a 1- charge for X and a 2- charge for Y.(a) Which combinations of these ions produce ionic compoundswhere there is a 1:1 ratio of cations and anions?(b) Among the combinations in part (a), which leads tothe ionic compound having the largest lattice energy?[Section 8.2]arrow_forwardIllustrated are four ions — A, B, X, and Y— showing their relativeionic radii. The ions shown in red carry positive charges: a2+ charge for A and a 1+ charge for B. Ions shown in blue carrynegative charges: a 1- charge for X and a 2- charge for Y.(a) Which combinations of these ions produce ionic compoundswhere there is a 1:1 ratio of cations and anions?(b) Among the combinations in part (a), which leads tothe ionic compound having the largest lattice energy?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
