
Concept explainers
a)
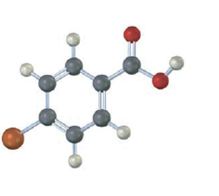
Interpretation:
Whether the
Concept introduction:
Electron releasing substituents, if present, in the benzene ring destabilize the carboxylate anion and makes the compound less acidic. On the other hand, electron withdrawing substituents, if present, in the benzene ring stabilize the carboxylate anion and makes the compound more acidic.
To state and explain:
Whether the carboxylic acid represented by the model is more acidic or less acidic than benzoic acid.
b)
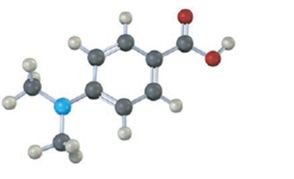
Interpretation:
Whether the carboxylic acid represented by the model is more acidic or less acidic than benzoic acid is to be stated and explained.
Concept introduction:
Electron releasing substituents, if present, in the benzene ring destabilize the carboxylate anion and makes the compound less acidic. On the other hand, electron withdrawing substituents, if present, in the benzene ring stabilize the carboxylate anion and makes the compound more acidic.
To state and explain:
Whether the carboxylic acid represented by the model is more acidic or less acidic than benzoic acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry
- Select the stronger acid from pair and explain your reasoning. For stronger acid, write a structural formula for its conjugate base. Q.) CH3CH2OH or CH3C=(three sticks in equals)CHarrow_forwardRank the following compounds in their correct order of acidity. 1=Most acidic and 4=least acidic.arrow_forwardb) Compounds in which the –OH of the carboxyl group is replaced by certain other groups are called carboxylic acid derivatives. The most important of which are acyl halides, acid anhydrides, esters and amides. Discuss the relationship between carboxylic acid, acid chloride and acid anhydridearrow_forward
- Please complete the following question. There is no need to provide an explaination. 1arrow_forward3) Consider the molecules of but-2-enal and but-3-enal below. Both molecules can be deprotonated by a strong base such as lithium diisopropylamide (LDA). H 2 H But-3-enal But-2-enal Which molecule is more acidic? Why? Use a reaction coordinate diagram(s) to explain your answer.arrow_forwardAlthough we normally think of acetic acid as an acid, it can also act as a weak base. Treated with a strong acid, it can become protonated. The conjugate acid of acetic acid is shown. H₂C :O-H H* Draw curved arrows for resonance structure 1. H₂C Derive a resonance structure for this ion which, taken with the given structure, would show that the two -OH groups are equivalent, the two C-O bonds are equivalent, and the positive charge is shared equally by the two oxygens. с The skeleton for the second resonance structure is provided. Add the bond(s), charge(s), and unshared pairs of electrons. Then draw the curved arrows to show the flow of electrons that transforms each resonance structure into the other. : OH || H3C- I.. -H OH :0 -H Complete the structure of resonance structure 2 and draw curved arrows to transform it to resonance structure 1. *K H₂C -C OH OHarrow_forward
- Consider the following compounds OH OH D A NH2 H E F The most basic compound is Choose... + The compound(s) that is (are) 2º alcohol Choose... + The compound(s) that is (are) considered as hydrocarbon Choose... The compound that has the strongest hydrogen-bonding is Choose...arrow_forwardArrange these compounds in order of increasing acidity: 2,4-dichlorophenol, phenol, cyclohexanol.arrow_forward2.) Draw the products of the following reactions. Provide an arrow-pushing mechanism for each reaction. Label the acid, base, conjugate base, and conjugate acid. Finally, indicate the direction of equilibrium. 요요 + NaHarrow_forward
- 2. It is known that phenol is unusually more acidic than the common alcohols. However, aniline is unusually less basic than the common amines. Explain the above facts with the help of resonance structures. • • NH2 : ОН Phenol Anilinearrow_forwardRank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.arrow_forwardWhy is ethanol least acidic?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
