
Concept explainers
Consider the photograph and illustrations of table salt. Do they include a model? Do they include a depiction of matter at the macroscopic, microscopic, and/or particulate levels? Explain your answers.
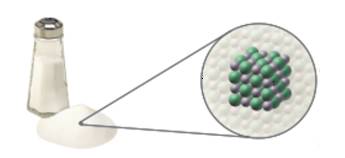
To predict the given photograph and illustrations of table salt, identify the depiction of matter at the macroscopic, microscopic, and/or particulate levels with explanation.
Interpretation:
Macroscopic, microscopic and particulate forms of matter are the three forms of matter.
Macroscopic can be viewed by the naked eyes, but we will be able to view microscopic form only by an optical microscope or electron microscope. Matter at the particulate level is so small that even with electron microscope it is really very difficult see them. Scientist use model to understand matter at particulate level. Models is representation of something
Concept introduction:
Answer to Problem 2.1TC
The art depicts a model of table salt at the particulate level. The photograph shows the macroscopic level
Explanation of Solution
Macroscopic, microscopic and particulate forms of matter are the three forms of matter.
Macroscopic can be viewed by the naked eyes, but we will be able to view microscopic form only by an optical microscope or electron microscope. Matter at the particulate level is so small that even with electron microscope it is really very difficult see them. Scientist use model to understand matter at particulate level. Models are representation of something. From the above statement we can say that the art depicts a model of table salt at the particulate level. The photograph shows at the macroscopic level
Thus the art depicts a model of table salt at the particulate level. The photograph shows the macroscopic level
Want to see more full solutions like this?
Chapter 2 Solutions
Introductory Chemistry: An Active Learning Approach
- Constant monitoring of acceptable concentrations of various dissolved substances is critical to keep our drinking water safe. In many areas, the drinking water standard for nitrate ions is 5.00 ppb. Calculate the mass, in micrograms, of nitrate ions allowed in one 254 mL glass of tap water. Record only your numerical answer with the correct number of significant digits.arrow_forwardAn alcoholic fermentation was conducted in the laboratory using 280 g glucose as substrate with a working volume of 2,000 mL. Assuming that the sugar is completely fermented to ethanol. Calculate the volume of ethyl alcohol in liters produced. The density of ethyl alcohol is 0.79 g/mL and its concentration or percent ethyl alcohol by volume produced assuming that no volume changed during fermentation.arrow_forward1. Seawater is composed of salt, sand, and water. Is seawater a heterogenous mixture or homogenous mixture? 2. Suppose there are four unknowns in bottles: three of the bottles are solutions and one is a pure substance. How are you going to determine which of the unknown is a pure substance?arrow_forward
- Imagine a world in which all the elements existed, but they couldn't form any chemical bonds with each other. What kinds of substances could exist in such a world? What kinds of substances could not exist? What do you think this world would be like?arrow_forwardRead the descriptions below of two substances and an experiment on each. Decide whether the result of the experiment tells you the substance is a pure substance or a mixture, if you can. • Sample A is 100. g of a coarse grey powder with a faint unpleasant smell. 15. g of the powder is put into a funnel lined with a sheet of thick paper. Distilled water is poured slowly over the powder. All of the powder disappears, and the water under the funnel turns a deep purple. • Sample B is a solid yellow cube with a total mass of 50.0 g. The cube is ground to a fine orange powder and added to a 500 mL beaker full of water. The beaker is stirred vigorously. Some of orange powder settles to the bottom of the beaker, and some rises to the top and floats on the water. When the powder at both the bottom and the top of the beaker is filtered out, dried, and weighed, the total mass is measured to be 50.1 g. pure substance Is sample A made from a pure substance or a mixture? If the description of the…arrow_forwardSoft wood chips weighing 17.2 kg are placed in an iron vessel and mixed with 150.1 kg water and 22.43 kg sodium hydroxide. A steel lid seals the vessel, which is then placed in an oven at 250°C for 6 hours. Much of the wood fiber decomposes under these conditions; the vessel and lid do not react.(a) Classify each of the materials mentioned as a substance or mixture. Subclassify the substances as elements or compounds.(b) Determine the mass of the contents of the iron vesselafter the reaction.arrow_forward
- Prepare a solution of NaCl by accurately weighing approximately 1.45g of NaCl into a 100 mL beaker / conical flask. Record the mass of the sample. Add 25 mL distilled water. Mix until all crystals dissolve. Prepare a solution of magnesium chloride by accurately weighing approximately 3.61 g of MgCl2٠6H20 into a 100 mL beaker / conical flask. Record the mass of the sample. Add 25 mLdistilled water. Mix until all the crystals dissolve. 1. Calculate the molality of the NaCℓ and MgCℓ2.6H2O solutions. 2. Calculate the total mass of water (solvent) in kg: a) Mass of crystal water added (kg): b) Mass of water added (kg): c) Total mass of water added (kg): 3. Calculate the molality of MgCℓ2. All questions follow on from each other. Thanks very much.arrow_forwardSuppose there are four unknowns in bottles: three of the bottles are solutions and one is a pure substance. How are you going to determine which of the unknown is a pure substance? Seawater is composed of salt, sand, and water. Is seawater a heterogenous mixture or homogenous mixture?arrow_forwardYou are working in the laboratory and are testing the salinity of ocean water in various areas along the gulf coast. What is the mass % of sodium chloride in a 250.0 mL sample that contains 6.80 g sodium chloride? Assume the density of the ocean water is 1.03g/mL.arrow_forward
- You are given a hammer a battery, a bulb, wires and switch(a) How could you use them to distinguish between samples of metals and non metals?(b) Assess the usefulness of these tests to distinguish between metals and non-metalsarrow_forwardThere is one mole of water in the beaker. A student measured the weight of the 1 mole of water of 18.02 g after using scale. Calculate the molar mass of water (H2O) in grams using your periodic table. [1 amu = 1.6606×10−24?1.6606×10−24g ]. How does your answer compare to the student's measurement? What can you conclude?arrow_forwardComplete the table. Table 1.2 Description of test samples. Sample Skeletal Structure Description (i.e., physical state, color) Cooking oil Acetic acid (vinegar) Sucrose (sugar) Monosodium glutamate (vetsin) Cellulose (cotton fiber) Isopropyl or ethyl alcohol Fatty acid salts (soap) Kerosene Naphthalene ballsarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




