
Concept explainers
(a)
Interpretation : To classify the given saccharide as mono, di, tri or polysaccharide.
Concept Introduction : Carbohydrates are composed of certain monomer units called as monosaccharides. Monosaccharides are polyhydroxy
(a)
Answer to Problem 75UTC
Melezitose is a tri-saccharide as it is composed of three monosaccharide units.
Explanation of Solution
Given:
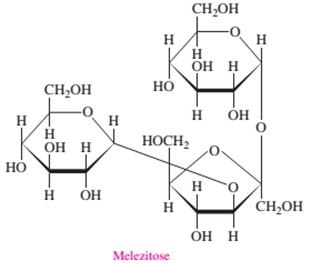
In the given structure of molecule there are three sugar molecules bonded with each other. Hence there are three monosaccharide units bonded to each other by two glycosidic linkages that make it a tri-saccharide molecule.
(b)
Interpretation : To identify the keto and aldohexose present in melezitose.
Concept Introduction : Carbohydrates are composed of certain monomer units called as monosaccharides. Monosaccharides are polyhydroxy aldehyde and ketones which are bonded with each other to form polysaccharides.
(b)
Answer to Problem 75UTC
Melezitose is composed of two aldohexose (glucose) and one ketohexose (fructose) units.
Explanation of Solution
In the given structure of molecule there are three sugar molecules; out of which two are pyranose rings with total 6 C atoms hence they should be aldohexose monosaccharides whereas one is furanose ring bonded with two −CH2OH groups at both terminals that proves it as a ketohexose. Hence Melezitose is composed of two aldohexose and one ketohexose units.
Want to see more full solutions like this?
Chapter 18 Solutions
Basic Chemistry
- (19, 9) Draw a structural formula for the major organic product of the reactions shown below. You do not have to consider stereochemistry.arrow_forward15.6 Stearic acid and linoleic acid each have 18 carbon atoms. Why does stearic acid melt at 69 °C but linoleic acid melts at -5 °C? 15.7 Draw the line-angle formula for each of the following fatty acids: a. palmitic acid b. oleic acid 15.8 Draw the line-angle formula for each of the following fatty acids: a. stearic acid b. linoleic acidarrow_forward23.71 a,b,c,darrow_forward
- C.14. What is the correlation between ketone levels with insulin value in a diabetic patient?arrow_forward16.48 Oxaloacetate is an inhibitor of succinate dehydrogenase. COO COO™ CH₂ CH₂ COO Succinate CH₂ C=O COO Oxaloacetate a. Would you expect oxaloacetate to be a competitive or a noncompetitive inhibitor? Why? b. Would oxaloacetate bind to the active site or elsewhere on the enzyme? c. How would you reverse the effect of the inhibitor?arrow_forward6) The disaccharide shown is a но но. HO OH но H. OH B) non reducing sugar A) reducing sugar 7) The glycosidic bond in the following structure can be described as a: OH но. но он OHI B) B (1.1) C) a (14) D) B (14) E) a (2,1) A) a (1,1)arrow_forward
- Draw the structure of the disaccharide with a systematic name of: (16)-B-D glucopyranosyl-B-D glucopyranosearrow_forward23.58: a,b,carrow_forward24.98 Draw the condensed structural formula for and give the name of the amino acid formed when the following a-keto acids undergo transamination with glutamate: (24.9) b. 0 || 70-C-CH₂-C-C-O CH3 0 || 0 0 |||| CH3-CH-CH2₂-C-C-Oarrow_forward
- Draw Hayworth projection for the disaccharide gentobiose given that the glycoside linkage is (16)arrow_forward13.10 Classify each of the following monosaccharides as an aldo- pentose, ketopentose, aldohexose, or ketohexose: a. A solution of xylose is given to test its absorption by the intestines. H H- -C-OH HO C-H HIC OH CH₂OH Xylose b. Tagatose, found in fruit, is similar in sweetness to sugar. CH₂OH C=0 HO C-H HO C-H HIC OH CH₂OH Tagatose 41/42 MOarrow_forward13.42 For each of the following, give the monosaccharide units produced by hydrolysis, the type of glycosidic bond, and the name of the disaccharide including a or B: a. CH2OH CH₂OH Н HO Н ОН Н Н ОН Н ____Н Н ОН Н Н ОН ОН Нarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





