
Concept explainers
What
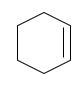
a.
(a)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
Starting alkene-
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction:
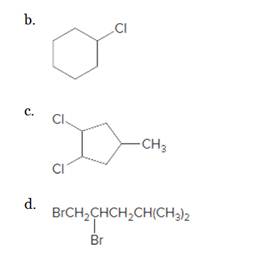
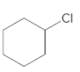
(b)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction;
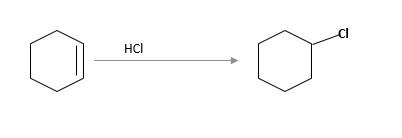
(c)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
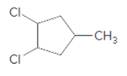
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
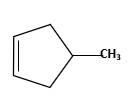 Starting alkene −
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction;
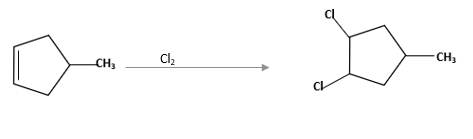
(d)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:

Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction:
Want to see more full solutions like this?
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
Additional Science Textbook Solutions
Basic Chemistry (5th Edition)
Inorganic Chemistry
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Principles of Chemistry: A Molecular Approach (3rd Edition)
Chemistry by OpenStax (2015-05-04)
- Name the given alkenes using systematic names. Consider alkene 1. alkene 1 name: H3C. CH2 .CH FCH2 H3C CH3 Consider alkene 2. alkene 2 name: H2 .C CH2 H2 CH3 Consider alkene 3. alkene 3 name: CH2 .CH2 H3C° IUarrow_forwardDraw the condensed structural formula or skeletal formula, if cyclic, for the alkene that is the major product from each of the following dehydration reactions: H* а. CHз — СН2— СH-— СH2— ОН Heat ОН ОН b. H+ с. H* Heat Нeatarrow_forwardwhat is the most stable alkene?arrow_forward
- explain Commercial Importance of Alkenesarrow_forwardCompounds with two carbonyl groups are named as alkane diones, for example: R 2,3-butanedione The compound above is an artificial flavor added to microwave popcorn and movie-theater popcorn to simulate the butter flavor. Interestingly, this very same compound is also known to contribute to body odor. Name the following compounds: cyclohexane-1,3-dione cycloheptane-1,4-dione gily nonane-2,8-dionearrow_forwardThis type of hydrocarbons contain at least one double or triple bond. Unsaturated hydrocarbons Saturated hydrocarbons Polyunsaturated hydrocarbons O Supersaturated hydrocarbons The most acidic among the hydrocarbons. Alkynes Alkenes Alkanes Arenes The number of secondary carbon(s) in this compound. CH3 CH,-C CHCH, CH3 CIarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning  World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





