
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.SE, Problem 42AP
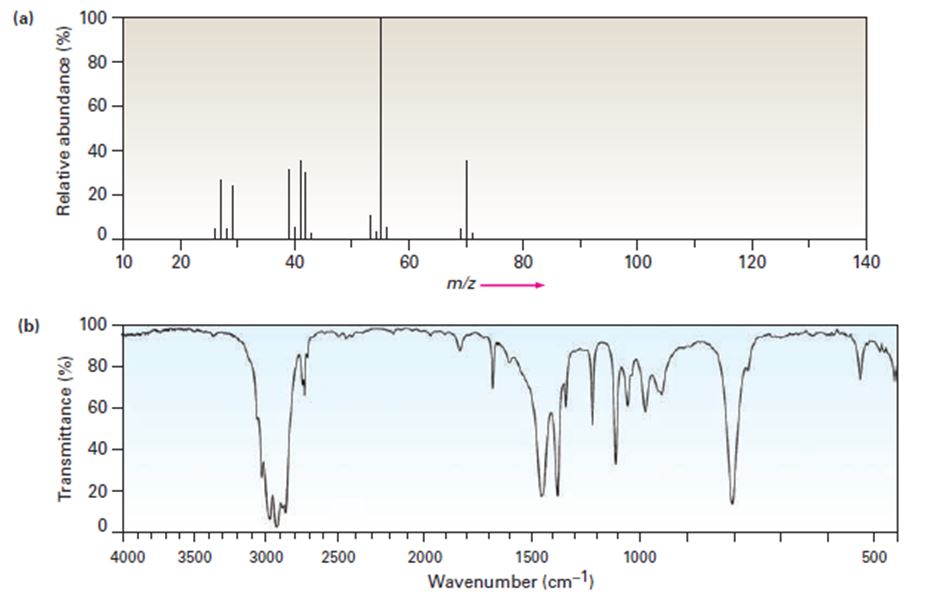
The mass spectrum (a) and the infrared spectrum (b) of another unknown hydrocarbon are shown. Propose as many structures as you can.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
How can you distinguish aldehydes, ketones, and carboxylic acids from each other using IR spectra? Explain using specific examples.
Shown below are the IR spectrum and the Mass Spectrum of a compound of molecular formula C3H8O. Using the spectral data, determine the structure of the compound. Provide evidence from both spectra to support your answer.
c) The mass spectrum of 2,3-dibromopentane (CH; CHBr CHBr CH₂ CH3) includes the following peaks.
Mass number (m/z) Relative abundance
15
29
107
109
199
201
203
i. What is the mass number of molecular ion, [CH; CHBr CHBr CH₂ CH₂]? Show your working.
ii. Identify the molecular formula (including isotopic composition where relevant) of the 6 peaks.
a
b
d
22
e
f
Mass number (m/z)
15
29
107
109
199
100
39
44
45
0.3
0.6
0.3
201
203
Molecule formula
[CH₂]
Chapter 12 Solutions
Organic Chemistry
Ch. 12.2 - Prob. 1PCh. 12.2 - Two mass spectra are shown in FIGURE 12-8. One...Ch. 12.3 - What are the masses of the charged fragments...Ch. 12.3 - Prob. 4PCh. 12.5 - Prob. 5PCh. 12.5 - Prob. 6PCh. 12.7 - What functional groups might the following...Ch. 12.7 - How might you use IR spectroscopy to distinguish...Ch. 12.8 - Prob. 9PCh. 12.8 - Where might the following compounds have IR...
Ch. 12.8 - Where might the following compound have IR...Ch. 12.SE - Prob. 12VCCh. 12.SE - Show the structures of the fragments you would...Ch. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - Write molecular formulas for compounds that show...Ch. 12.SE - Camphor, a saturated monoketone from the Asian...Ch. 12.SE - The nitrogen rule of mass spectrometry says that a...Ch. 12.SE - In light of the nitrogen rule mentioned in Problem...Ch. 12.SE - Nicotine is a diamino compound isolated from dried...Ch. 12.SE - The hormone cortisone contains C, H, and O, and...Ch. 12.SE - Halogenated compounds are particularly easy to...Ch. 12.SE - Prob. 22APCh. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - 2-Methylpentane (C6H14) has the mass spectrum...Ch. 12.SE - Assume that you are in a laboratory carrying out...Ch. 12.SE - What fragments might you expect in the mass...Ch. 12.SE - How might you use IR spectroscopy to distinguish...Ch. 12.SE - Would you expect two enantiomers such as...Ch. 12.SE - Would you expect two diastereomers such as meso-2,...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - How could you use infrared spectroscopy to...Ch. 12.SE - Prob. 32APCh. 12.SE - At what approximate positions might the following...Ch. 12.SE - How would you use infrared spectroscopy to...Ch. 12.SE - At what approximate positions might the following...Ch. 12.SE - Assume that you are carrying out the dehydration...Ch. 12.SE - Assume that you are carrying out the base-induced...Ch. 12.SE - Prob. 38APCh. 12.SE - Carvone is an unsaturated ketone responsible for...Ch. 12.SE - Prob. 40APCh. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - 4-Methyl-2-pentanone and 3-methylpentanal are...Ch. 12.SE - Grignard reagents undergo a general and very...Ch. 12.SE - Ketones undergo a reduction when treated with...Ch. 12.SE - Nitriles, R–=C≡N, undergo a hydrolysis...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - Prob. 50AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A mass peak at m/z = 59 appears in the mass spectrum of an amide, C5H11NO. Draw the structure of a molecule that is consistent with this result.arrow_forwardThe base peak in the mass spectrum of an alkane, C7H16, appears at m/z =57. Draw a molecule that is consistent with these data.arrow_forward. Deduce the structure of an unknown compound with the molecular formula of C5H₁2O using the information given by its infrared spectrum. ANSWER A) НО. B) D) Intensity Frequency (peak): (cm): 3300 m EEEEE m m m m m 2900 2800 1465 1450 1375arrow_forward
- 2. In a mass spectrum of a compound, the following data (intensity) is given. M+ = 80, M+ +1 = 8.8, M+ +2 = 26. This compound will have carbons and will contain a a. 8, sulfur b. 9, silicon c. 10, chlorine d. 11, brominearrow_forwardA compound containing only carbon, nitrogen, oxygen, and hydrogen contains four carbon atoms. If the M+ peak in its mass spectrum appears at m/z = 87, then how many nitrogen atoms does it contain?arrow_forwardFollowing is the mass spectrum of an unknown compound. The two highest peaks are at mlz 120 and 122. Suggest a structure for this compound. (Data from http://webbook .nist.gov/chemistry/.) 100 41 80 20 120 122 0 rt 10 20 30 40 60 70 110 140 80 90 m/z 50 100 120 130 150 160 Relative Abundancearrow_forward
- Which atom do you expect is present in this spectrum? 100 Rel. Intensity 80 60 60 40 40 20 20 0.0 0.0 15 30 45 60 75 90 m/z NIST Chemistry WebBook (https://webbook.nist.gov/chemistry) Only hydrocarbons Bromine Chlorine O Nitrogenarrow_forwardThe mass spectrum of an alkene, C8H16, exhibits a peak at m/z = 41. Draw two isomers that are consistent with these data.arrow_forwardAter analyzing a mass spectrum you conclude that the molecule being analyzed has the molecular formula C4H8; however, there are five possible constitutional isomers with that same molecular formula. the only thing that stands out in the mass spectrum is a strong fragment signal at m/z=41. Determine the possible constitutional isomers for C4H8 and explain which isomer matches the mass spectrum.arrow_forward
- This is an infrared spectra of a compound whose molar mass is 108 g/mol. Determine the molecular formula and propose a structure.arrow_forwardLabel the molecular ion, the base peak, and the M + 1 peak in the mass spectrum of pentane (C5H12).arrow_forward1. Given the parent compound, chlorobenzene, draw the mass spectrum fragment that is observed at m/z 77. Include any hydrogen atoms and the charge. H H C H H H 2. Given the parent compound, isobutyl-benzene, draw the mass spectrum fragment that is observed at m/z 91. Include any hydrogen atoms and the charge. CH3 CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY