
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN: 9781305580343
Author: Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.21QP
Best Lewis Formula and Molecular Geometry
A student writes the Lewis electron-dot formula for the carbonate anion, CO32−, as
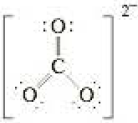
- a Does this Lewis formula obey the octet rule? Explain. What are the formal charges on the atoms? Try describing the bonding for this formula in valence bond terms. Do you have any difficulty doing this?
- b Does this Lewis formula give a reasonable description of the electron structure, or is there a better one? If there is a better Lewis formula, write it down and explain why it is better.
- c The same student writes the following resonance description for CO2:
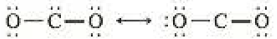
Is there something wrong with this description? (What would you predict as the geometries of these formulas?)
- d Is one or the other formula a better description? Could a value for the dipole moment help you decide?
- e Can you write a Lewis formula that gives an even better description of CO2? Explain your answer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part 4:
4a. Draw the best Lewis structure for the CH3COOH (acetic acid) molecule. (Hint: the acidic
H is bonded to O in an oxy-acid molecule).
Evaluate each atom using formal charge to prove you have the best structure. Show your
math work as well as your formal charge answers.
4b. Draw all the resonance structures for the CH3COO- (acetate ion). (Hint: make sure you
have the best initial structure first!)
Draw a Lewis structure of your choice that has at least three equally reasonable resonance structures. You must show your work as to how the structure was drawn including showing valence electrons, any structures you draw that don’t work and require you to show a multiple bond, and formal charges on each atom that has a formal charge. For example, Nitrate ion has three structures (but you can’t draw this one). All three structures have formal charges and no one structure is preferred based on formal charge. Carbon dioxide would not be a valid choice. While it does have three structures, the structure that has two double bonds is the preferred structure while the structures with triple bonds are not equivalent to the double bonded version as they have multiple formal charges present.
A resonance hybrid is a structure that can be depicted by more than one valid Lewis structure.
part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.
part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.
part3: Draw the least important resonance contributor for fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized and should include all nonzero formal charges and all nonbonding electrons.
Chapter 10 Solutions
General Chemistry - Standalone book (MindTap Course List)
Ch. 10.1 - An atom in a molecule is surrounded by four pairs...Ch. 10.1 - Use the VSEPR method to predict the geometry of...Ch. 10.1 - Prob. 10.2ECh. 10.2 - Bromine trifluoride, BrF3, has a nonzero dipole...Ch. 10.2 - Which of the following would be expected to have a...Ch. 10.2 - Two molecules, each with the general formula AX3,...Ch. 10.3 - Using hybrid orbitals, describe the bonding in NH3...Ch. 10.4 - Describe the bonding on the carbon atom in carbon...Ch. 10.4 - Dinitrogen difluoride (see Example 10.5) exists as...Ch. 10.4 - Prob. 10.3CC
Ch. 10.6 - The C2 molecule exists in the vapor phase over...Ch. 10.6 - Give the orbital diagram and electron...Ch. 10 - Describe the main features of the VSEPR model.Ch. 10 - According to the VSEPR model, what are the...Ch. 10 - Why is a lone pair expected to occupy an...Ch. 10 - Prob. 10.4QPCh. 10 - Explain why nitrogen trifluoride has a small...Ch. 10 - Prob. 10.6QPCh. 10 - What is the angle between two sp3 hybrid orbitals?Ch. 10 - Prob. 10.8QPCh. 10 - Prob. 10.9QPCh. 10 - How does the valence bond description of a...Ch. 10 - Prob. 10.11QPCh. 10 - What factors determine the strength of interaction...Ch. 10 - Prob. 10.13QPCh. 10 - Prob. 10.14QPCh. 10 - Prob. 10.15QPCh. 10 - Describe the bonding in O3, using molecular...Ch. 10 - Prob. 10.17QPCh. 10 - Which of the following molecular geometries does...Ch. 10 - Which of the following would be a polar molecule?...Ch. 10 - Prob. 10.20QPCh. 10 - Best Lewis Formula and Molecular Geometry A...Ch. 10 - Prob. 10.22QPCh. 10 - Prob. 10.23QPCh. 10 - Which of the following molecular models correctly...Ch. 10 - Prob. 10.25QPCh. 10 - Prob. 10.26QPCh. 10 - Indicate what hybrid orbital depicted below is...Ch. 10 - An atom in a molecule has two bonds to other atoms...Ch. 10 - Two compounds have the same molecular formula,...Ch. 10 - A neutral molecule is identified as a...Ch. 10 - Acetic acid, the sour constituent of vinegar, has...Ch. 10 - What are the bond angles predicted by the VSEPR...Ch. 10 - Predict the shape or geometry of the following...Ch. 10 - Use the electron-pair repulsion model to predict...Ch. 10 - Predict the geometry of the following ions, using...Ch. 10 - Use the VSEPR model to predict the geometry of the...Ch. 10 - For each of the following molecules, state the...Ch. 10 - For each of the following molecules, state the...Ch. 10 - Prob. 10.39QPCh. 10 - From the electron-pair repulsion model, predict...Ch. 10 - Predict the geometries of the following ions,...Ch. 10 - Name the geometries expected for the following...Ch. 10 - a The molecule AsF3 has a dipole moment of 2.59 D....Ch. 10 - a The molecule BrF3 has a dipole moment of 1.19 D....Ch. 10 - Which of the following molecules would be expected...Ch. 10 - Which of the following molecules would be expected...Ch. 10 - What hybrid orbitals would be expected for the...Ch. 10 - What hybrid orbitals would be expected for the...Ch. 10 - What hybrid orbitals would be expected for the...Ch. 10 - What hybrid orbitals would be expected for the...Ch. 10 - a Mercury(II) chloride dissolves in water to give...Ch. 10 - a Nitrogen trifluoride, NF3, is a relatively...Ch. 10 - a Carbonyl fluoride, COF2, is an extremely...Ch. 10 - a The molecule HNNH exists as a transient species...Ch. 10 - The hyponitrite ion, ONNO, exists in solid...Ch. 10 - Fumaric acid, C4H4O4, occurs in the metabolism of...Ch. 10 - Describe the electronic structure of each of the...Ch. 10 - Use molecular orbital theory to describe the...Ch. 10 - Prob. 10.59QPCh. 10 - Write the molecular orbital configuration of the...Ch. 10 - Predict the molecular geometry of the following: a...Ch. 10 - Prob. 10.62QPCh. 10 - Which of the following molecules or ions are...Ch. 10 - Which of the following molecules or ions are...Ch. 10 - Describe the hybrid orbitals used by each carbon...Ch. 10 - Prob. 10.66QPCh. 10 - Explain how the dipole moment could be used to...Ch. 10 - Two compounds have the formula Pt(NH3)2Cl2....Ch. 10 - Explain in terms of bonding theory why all four...Ch. 10 - Explain in terms of bonding theory why all atoms...Ch. 10 - What is the molecular orbital configuration of...Ch. 10 - Prob. 10.72QPCh. 10 - Calcium carbide, CaC2, consists of Ca2+ and C22...Ch. 10 - Sodium peroxide, Na2O2, consists of Na+ and O22...Ch. 10 - The oxygen oxygen bond in O2 is 112 pm and in O2...Ch. 10 - The nitrogennitrogen bond distance in N2 is 109...Ch. 10 - Using molecular orbital theory, determine the...Ch. 10 - The ionization energy of O2 is smaller than the...Ch. 10 - Prob. 10.79QPCh. 10 - Prob. 10.80QPCh. 10 - Prob. 10.81QPCh. 10 - Prob. 10.82QPCh. 10 - What is the biological importance of stratospheric...Ch. 10 - Prob. 10.84QPCh. 10 - Prob. 10.85QPCh. 10 - The bond length in C2 is 131 pm. Compare this with...Ch. 10 - Calcium carbide, CaC2, has an ionic structure with...Ch. 10 - Write Lewis formulas for the BF molecule (two with...Ch. 10 - Boron trifluoride, BF3, reacts with ammonia, NH3,...Ch. 10 - Prob. 10.90QPCh. 10 - Allene (1,2-propadicne), a gas, has the following...Ch. 10 - Prob. 10.92QPCh. 10 - The triiodide ion, I3, and the azide ion, N3, have...Ch. 10 - Hydrogen azide (also known as hydrazoic acid),...Ch. 10 - Prob. 10.95QPCh. 10 - A molecule XF6 (having no lone pairs) has a dipole...Ch. 10 - Describe the molecular orbital configurations of...Ch. 10 - Prob. 10.98QPCh. 10 - Three different compounds have the same molecular...Ch. 10 - Prob. 10.100QPCh. 10 - Prob. 10.101QPCh. 10 - Solid sulfur normally consists of crystals of S8...Ch. 10 - Prob. 10.103QPCh. 10 - Consider the bonding in nitrate ion, NO3. First...Ch. 10 - A molecular compound is composed of 52.5% Xe,...Ch. 10 - A molecular compound is composed of 58.8% Xe,...Ch. 10 - A compound of chlorine and fluorine. ClFn, reacts...Ch. 10 - Excess fluorine, F2(g), reacts at 150C with...Ch. 10 - Prob. 10.109QPCh. 10 - One resonance formula of benzene, C6H6, is What is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.arrow_forwardAn incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of zero. H Н—N—С- -о—с—Н H H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges. Click and drag to start drawing a structure.arrow_forwardConsider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. (IMAGE WITH POSSIBILITIES) h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?arrow_forward
- Consider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C? please answer c, d and earrow_forwardConsider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?arrow_forwardCompare the average N–O bond in your Lewis structures of NO2+ and NO2–. Based on the trends you identified above, which average N–O bond do you expect to be stronger? Which do you expect to be shorter? Explain your reasoning. Stronger N–O Bond NO2+ NO2– Shorter N–O Bond NO2+ NO2–arrow_forward
- Choose the best Lewis structure from those presented below. (It will help to work out the missing formal charges for the atoms in these different structures) [Image description: Lewis structure A has a N atom singly bound to two O atoms. There is one electron pair on N. One O atom has three electron pairs and the other O atom has two electron pairs and another single bond to a H atom. Lewis structure B has a O atom, 01, singly bound to a N atom and doubly bound to another O atom, O2. There is one electron pair on O1 and two electron pairs on O2. On the N atom there are two electron pairs and another single bond to a H atom. Lewis structure C has a O atom, 01, singly bound to another O atom, O2, and doubly bound to a N atom. There are is one electron pair on 01 and two electron pairs plus another singly bond to a H atom on 02. There are two electron pair on N. Lewis structure D has a N atom singly bound to a O atom, O1, and doubly bound to another O atom, O2. There are two electron…arrow_forwardHow do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?arrow_forwardConsider the formate ion, HCO2-, which is the anion formed when formic acid lose an H+ ion. The H and the two O atoms are bonded to the central C atom. a) Draw the best Lewis structure for this ion. b) Are resonance structures needed to describe this structure? c) Would you predict that the C-O bond lengths in the formate ion would be longer or shorter relative to those in CO2?arrow_forward
- An incomplete Lewis structure is shown below. The structure only shows the atoms and how they are connected. The molecule has a net charge of +1. H H N H Complete the Lewis structure giving all atoms full octets. If there is more than one way to do this, draw resonance structures showing all possibilities. If not, just draw one Lewis structure. Be sure to write in any non-zero formal charges. Click and drag to start drawing a structure. olo Ar 6arrow_forwardWhat possible error(s) exist in the Lewis structure (assume we are trying to represent the best possible Lewis structure for the NO₂S ion knowing N is the central atom in this polyatomic ion)? [:ö==S: N= CO :O: The best structure would have double bond and two lone pairs on each oxygen atom and a single bond with three lone pairs on the sulfur. There are no errors. This is the best possible structure. The Lewis structure above does not minimize formal charges, thus is the not the best possible structure. The nitrogen atom has an expanded octet, and this structure is impossible. The Lewis structure contains the wrong number of electrons, thus this structure is impossible.arrow_forwardDraw two valid Lewis structures for POCl. Structure A should have Phosphorus as the central atom (O-P- Cl) and Structure B should have Oxygen as the central atom (P-O-Cl). In both structures, the Cl atom should be singly bonded to the central atom What is the formal charge of EACH atom is Structure A? What is the formal charge of EACH atom is Structure B? On the basis of formal charge, which is the best structure for this compound? Explain why you chose that partcular structure.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Brooks Cole
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY