
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
11th Edition
ISBN: 9780134193601
Author: Petrucci
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 1, Problem 94FP
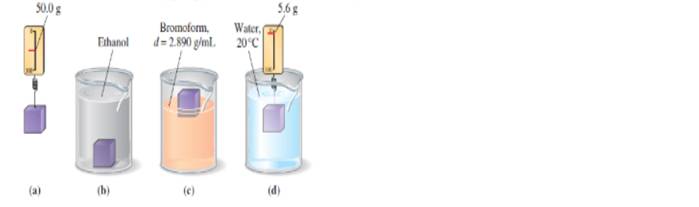
The accompanying sketches suggest four observations made on a small block of plastic material. Tell what conclusions can be drawn from each sketch, and conclude by giving your best estimate of the density of the plastic.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A movie stunt performer is filming a scene where he swings across a river on a vine. The safety crew must use a vine with enough strength so that it doesn't
break while swinging. The stunt performer's mass is 88.0 kg, the vine is 12.0 m long, and the speed of the stunt performer at the bottom of the swing has been
determined to be 8.60 m/s. What is the minimum tension force (in N) the vine must be able to support without breaking?
N
Need Help?
Submit Answer
Read It
Provide a molecular interpretation for the observation that the viscosity
of a gas increases with temperature, whereas the viscosity of a liquid decreases
with increasing temperature
One of the laser commonly used to produce blue and green light in laser light shows in the Ar+ ion laser, which operates using gaseous Ar+ ions. Consider a gas of these Ar+ ions. Would you expect the compressibility factor for this system to be greater than, less than, or equal to 1? Explain your reasoning.
Chapter 1 Solutions
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
Ch. 1 - What are the principal reasons that one theory...Ch. 1 - Prob. 2ECh. 1 - A common belief among scientists is that there...Ch. 1 - Describe several ways in which a scientific law...Ch. 1 - Describe the necessary characteristics of an...Ch. 1 - Describe the necessary characteristics of a...Ch. 1 - State whether the following properties of matter...Ch. 1 - State whether the following properties are...Ch. 1 - Indicate whether each sample of matter listed is 8...Ch. 1 - Indicate whether each sample of matter listed is...
Ch. 1 - Suggest physical changes by which the following...Ch. 1 - What type of changephysical or chemicalis...Ch. 1 - Express each number in exponential notation....Ch. 1 - Express each number common decimal form. a....Ch. 1 - Express each value in exponential form. Where...Ch. 1 - Express each value in exponential form. Where...Ch. 1 - Indicate whether each of the following is an exact...Ch. 1 - Indicate whether each of the following is an exact...Ch. 1 - Prob. 19ECh. 1 - How many significant figures are shown in each of...Ch. 1 - Perform the following calculations; express each...Ch. 1 - Perform the following calculations; express each...Ch. 1 - Perform the following calculations and retain the...Ch. 1 - Express the result of each of the following...Ch. 1 - An American press release describing the 1986...Ch. 1 - Prob. 26ECh. 1 - Perform the following conversions. a. 0.127L=mL b....Ch. 1 - Prob. 28ECh. 1 - Perform the following from non-SI to SI units....Ch. 1 - Prob. 30ECh. 1 - Which is the greater mass, 3245 (g or 0.00515 mg?...Ch. 1 - Which is the greater mass, 3257 mg or 0.000475 kg?...Ch. 1 - The non-SI unit, hand (used by equestrians), is 4...Ch. 1 - The und furlong is used in horse racing. The unis...Ch. 1 - A sprinter runs the 100 yd dash in 9.3 s. At this...Ch. 1 - A non-SI unit of mass used in Pharmaceutical work...Ch. 1 - Prob. 37ECh. 1 - In an engineering reference book, you find that...Ch. 1 - Prob. 39ECh. 1 - The volumeofaredbloodcell isabout 90.010-12cm3 ....Ch. 1 - We want tomark offathermometer in it Celsius and...Ch. 1 - The highestandlowesttemperatures on record for San...Ch. 1 - The absolute zero of temperature is -273.15C....Ch. 1 - Prob. 44ECh. 1 - Prob. 45ECh. 1 - Prob. 46ECh. 1 - A 2.18 L sample of butyric acid, a substance...Ch. 1 - A 15.2 L sample of chloroform at 20 C has a mass...Ch. 1 - To determine the density of acetone, a 55.0 gal...Ch. 1 - To determine the volume of an irregularly shaped...Ch. 1 - A solution consisting of 8.50% acetone and 91.5%...Ch. 1 - Prob. 52ECh. 1 - A fertilizer contains 21% nitrogen by mass. What...Ch. 1 - A sample is found to have a density of 1.006 g/mL,...Ch. 1 - Prob. 55ECh. 1 - Calculate the mass of a cylinder of stainless...Ch. 1 - The densities are given at 20 C: water, 0.998g/cm3...Ch. 1 - To determine the approximate mass of a small...Ch. 1 - The density of aluminum is 2.70 g/ cm3. A square...Ch. 1 - The angle iron pictured here is made of steel with...Ch. 1 - In normal blood, there are about 5.4109 red blood...Ch. 1 - A technique once used by geologists to measure the...Ch. 1 - In a class of 76 students, the results of...Ch. 1 - A class of 84 students had a final grade...Ch. 1 - Prob. 65ECh. 1 - A solution containing 12.0% sodium hydroxide by...Ch. 1 - According to the rules on significant figures, the...Ch. 1 - Prob. 68IAECh. 1 - A solution used to chlorinate a home swimming pool...Ch. 1 - A standard 1.000 kg mass is to be tut from a bar...Ch. 1 - Prob. 71IAECh. 1 - Prob. 72IAECh. 1 - Magnesium occurs in seawater to the extent of 1.4...Ch. 1 - A typical rate of deposit of dust ("dustfall")...Ch. 1 - In the United States, the volume of irrigation...Ch. 1 - A Fahrenheit and a Celsius thermometer are...Ch. 1 - The accompanying illustration shows e 100.0 mL...Ch. 1 - Prob. 78IAECh. 1 - Prob. 79IAECh. 1 - A pycnometer (see Exercise 78) weighs 25.60 g...Ch. 1 - The Greater Vancouver Regional District (GVRO)...Ch. 1 - A Boeing 767 due to fly from Montreal to Edmonton...Ch. 1 - The following equation can be used to relate the...Ch. 1 - Prob. 84IAECh. 1 - A tabulation of datalists the following equation...Ch. 1 - The total volume of ice in the Antarctic is about...Ch. 1 - An empty 3.00 L bottle weighs 1.70 kg. Fled with...Ch. 1 - The filament in an incandescent light bulb is made...Ch. 1 - Blood alcohol content (BAC) is sometimes reported...Ch. 1 - In an attempt to determine any possible...Ch. 1 - Prob. 91FPCh. 1 - Prob. 92FPCh. 1 - The canoe gliding gracefully along the water in...Ch. 1 - The accompanying sketches suggest four...Ch. 1 - As mentioned on page 13, the MCO was lost because...Ch. 1 - In your own words, define or explain the following...Ch. 1 - Prob. 97SAECh. 1 - Explain the important distinctions between each...Ch. 1 - A procedure designed to test the truth or the...Ch. 1 - The fact that the volume of a fixed amount of gas...Ch. 1 - If a sample of matter cannotbe separated by...Ch. 1 - A good example of a homogeneous mixture is a. a...Ch. 1 - Compared withits mass on Earth, the mass of the...Ch. 1 - Which answer has the correct number of significant...Ch. 1 - Which two of the following masses are expressed to...Ch. 1 - Prob. 106SAECh. 1 - Prob. 107SAECh. 1 - Prob. 108SAECh. 1 - The density of water is 0.9982 g/cm2 at 20C....Ch. 1 - Two students each made four measurements of the...Ch. 1 - Prob. 111SAECh. 1 - List the blowing the order of increasing...Ch. 1 - Without doing detailed calculations, explain which...Ch. 1 - Prob. 114SAECh. 1 - Water, acompound, is a substance. Is there any...Ch. 1 - In the production of ammonia, the...Ch. 1 - Appendix E descries a useful study aid known as...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The density of liquid NH3 is 0.64 g/mL; the density of gaseous NH3 at STP is 0.0007 g/mL. Explain the difference between the densities of these two phases.arrow_forwardDraw and describe each of the three main phases of matter (solid, liquid, and gas) such that it is clear what the (i) spacing, (ii) order and (iii) speed of particles are in this phase. Pictures and descriptions are covered in the lecture outline. Your picture of the gas phase must include a measurement with a ruler of both the gas particle and the distance between particles.arrow_forwardWhat error would be introduced into the determination of the density of a solid object, if air bubbles were introduced into the liquid while measuring its volume by liquid displacement? Would the calculated density be higher or lower than its true value? Explain.arrow_forward
- Describe the process occurring at the molecular level thataccounts for the property of viscosity.arrow_forwardThe masses of benzene and an unknown liquid droplets falling off from the same stalagmometer at 20 ° C were measured as 0.052 g and 0.042 g, respectively. Since the surface tension of benzene at the test temperature is 28.9 dyn / cm, find the surface tension of the other liquid at the same temperature.arrow_forwardThe viscosity of carbon dioxide gas at 1 atm is 436 µP at 850°C. Calculate the apparent hard sphere of carbon dioxide at this temperature.arrow_forward
- The role of single molecule in nano particle junctionsarrow_forwardcl (aq) + NO₂ (aq) → NO(g) + Ch (g) Express your answer as a chemical equation. Identify all of the phases in your answer. Teamplang Symbole undo' regio ese keyboard shortcuts Help V 6C1₂(aq) + 2NO, (aq) + 8H* (g)→3C1₂(g) + 2NO(g) + 4H₂O A chemical reaction does not occur for this question.arrow_forwardFor each of the following statements, insert the letter T for True and Ffor false. Viscosity is a result of inter-molecular forces. The size and/or shape of the molecules will influence the viscosity. Long molecules that can become entangled will have a lower viscosity than a spherical molecule. The viscosity of a fluid increases with temperature.arrow_forward
- my topic is "Hazard placards on compressed gases For this assignment, you will be working in a small group to research the scenario indicated in the title of your group and explain how it is dependent on the properties of gases that you learned about in this module. You will compile your research in a short wiki page. The expectations for your group are as follows: • Each group member will research and contribute one source related to the application of gaseous properties to your assigned topic.arrow_forwardFind the magnitude and direction of the resultant force for the system of forces shown in the figure. Where F1 =20 N, F2 =5N, F3 =30 N, F4 =10N, F5 = 60N & F6 =70N. The angles 02 =15º, 03 =20° , 04 = 20°, 06 = 30 ° (ENTER ONLY THE VALUES IN THE BOXES BY REFERRING THE UNIT GIVEN IN BRACKET. ALSO UPLOAD YOUR HANDWRITTEN ANSWERS IN THE LINK PROVIDED) F2 F3 F4 F1 AF6 F5 (1) The horizontal component 2H of the given system (unit is in N) = (2) The horizontal component ZV of the given system (unit is in N) = (3) The resultant of the given force system is (unit in N) = (4) The direction of Resultant is =arrow_forwardA perfectly elastic 170L balloon is heated from 25.0°C to 62.3°C. To what volume (L) will it expand?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,
Viscosity, Cohesive and Adhesive Forces, Surface Tension, and Capillary Action; Author: Professor Dave Explains;https://www.youtube.com/watch?v=P_jQ1B9UwpU;License: Standard YouTube License, CC-BY