
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.SE, Problem 49AP
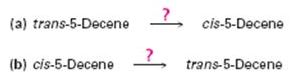
Occasionally, a chemist might need to invert the stereochemistry of an
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Consider a reaction where cis-but-2-ene is treated with a peroxy acid followed by OH- /H20. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.
Draw the structure for an alkene that gives the following reaction product.
1. Hg(OАc)2, Н2О
?
-CHCH3
2. NABH4
ОН
Ignore alkene stereochemistry.
You do not have to explicitly draw H atoms.
In cases where there is more than one answer, just draw one.
Visited
opy aste
ChemDoodle
Draw the alkene that would react with the reagent given to account for the product formed.
? +
+
H₂O
****
H₂S04
• You do not have to consider
stereochemistry.
• You do not have to explicitly draw H atoms.
• In cases where there is more than one answer, just draw one.
CH3
CH3 CHCCH3
| |
OH CH3
+1
Chapter 9 Solutions
Organic Chemistry
Ch. 9.1 - Prob. 1PCh. 9.1 - Prob. 2PCh. 9.3 - What products would you expect from the following...Ch. 9.4 - Prob. 4PCh. 9.4 - Prob. 5PCh. 9.4 - Prob. 6PCh. 9.4 - Prob. 7PCh. 9.5 - Using any alkyne needed, how would you prepare the...Ch. 9.7 - The pKa of acetone, CH3COCH3, is 19.3. Which of...Ch. 9.8 - Prob. 10P
Ch. 9.8 - Prob. 11PCh. 9.9 - Show the terminal alkyne and alkyl halide from...Ch. 9.9 - Beginning with acetylene and any alkyl halide...Ch. 9.SE - Name the following alkynes, and predict the...Ch. 9.SE - From what alkyne might each of the following...Ch. 9.SE - Prob. 16VCCh. 9.SE - The following cycloalkyne is too unstable to...Ch. 9.SE - Prob. 18MPCh. 9.SE - Assuming that strong acids add to alkynes in the...Ch. 9.SE - Prob. 20MPCh. 9.SE - Prob. 21MPCh. 9.SE - Prob. 22MPCh. 9.SE - Prob. 23MPCh. 9.SE - Prob. 24MPCh. 9.SE - Reaction of acetone with D3O+ yields...Ch. 9.SE - Give IUPAC names for the following compounds:Ch. 9.SE - Draw structures corresponding to the following...Ch. 9.SE - Prob. 28APCh. 9.SE - Prob. 29APCh. 9.SE - Prob. 30APCh. 9.SE - Predict the products from reaction of l-hexyne...Ch. 9.SE - Prob. 32APCh. 9.SE - Prob. 33APCh. 9.SE - Propose structures for hydrocarbons that give the...Ch. 9.SE - Identify the reagents a-c in the following scheme:Ch. 9.SE - Prob. 36APCh. 9.SE - Prob. 37APCh. 9.SE - Prob. 38APCh. 9.SE - How would you carry out the following...Ch. 9.SE - Prob. 40APCh. 9.SE - Synthesize the following compounds using 1-butyne...Ch. 9.SE - Prob. 42APCh. 9.SE - Prob. 43APCh. 9.SE - Prob. 44APCh. 9.SE - Prob. 45APCh. 9.SE - A hydrocarbon of unknown structure has the formula...Ch. 9.SE - Compound A(C9H12) absorbed 3 equivalents of H2 on...Ch. 9.SE - Hydrocarbon A has the formula C12H8. It absorbs 8...Ch. 9.SE - Occasionally, a chemist might need to invert the...Ch. 9.SE - Prob. 50APCh. 9.SE - Prob. 51APCh. 9.SE - Prob. 52APCh. 9.SE - Prob. 53APCh. 9.SE - Prob. 54APCh. 9.SE - Prob. 55APCh. 9.SE - Prob. 56APCh. 9.SE - Prob. 57AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 4. What are the products obtained from the following elimination reaction? Indicate the major product. CH;CH2CCH3 H2O CI 5. a) Determine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. CH;CH,CH,CH,CCH3 Br b) For the major elimination product obtained in 5a), which stereoisomer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forwardAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forwardAcyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. CI H₂N OH • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • Do not include counter-ions, e.g., Na+, I, in your answer. • In cases where there is more than one answer, just draw one. Sn [Farrow_forward
- 4. What are the products obtained from the following elimination reaction? Indicate the major product. CH3 CH,CH,ČCH, H2O 5. a) Determine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. CH;CH,CH,CH,CCH, Br b) For the major elimination product obtained in 5a), which stereoisomer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forwardProvide detailed step-wise mechanism for the following reaction. Be sure to show all intermediates, formal charges, and show the movement of electrons with curved arrows. a 4+H₂Q 044 OHarrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forward
- CH3 CH3 Br- Br2 .CH3 CH2Cl2 CH3 H3C H3C Br Electrophilic addition of bromine, Brɔ, to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH,Cl,. In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is formed. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions CH3 CH3 CH3 CH3 H3C H3C :Br: :Br:arrow_forwardDraw a structural formula for the major product of the reaction shown. H3C Cl2 H3C H20 Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer. opy aste - [F ChemDoodle Previous Nexarrow_forwardWhen treated with NaOH, the bromide below gives an alkene by the E2 mechanism, by eliminationof the circled atoms: (a) Draw the Newman projection from which elimination takes place. (b) Draw the mechanism. (c) Draw the product with the proper stereochemistry. (d) Assign the proper stereochemical descriptor to the product. (Z, E? Trans,CiS?) (e)What would happen to the stereochemistry of the product if the enantiomer of the starting material were used inthe reaction?arrow_forward
- Consider the following mechanism of reaction: CH3 CH, CH3 CH;C CH, + H-CI CH;CCH, + C: → CH;CCH, tert-butyl cation CI tert-butyl chloride In this reaction: The alkene and the carbocation intermediate are both nucleophilic HCl and CI" are both nucleophilic The alkene and HCI are both electrophilic The carbocation and CI" are both electrophilic The alkene and CI" are both nucleophilic AMeving to another question will save this responsearrow_forwardDraw the most stable resonance form for the intermediate in the following electrophilic substitution reaction. CH3 CH3 H3PO4 HO, + H3C CH3 HO HO First stage in synthesis of the epoxy and polycarbonate ingredient bisphenol-A You do not have to consider stereochemistry. Include all valence lone pairs in your answer. In cases where there is more than one answer, just draw one.arrow_forward9) Draw the conformer structure, Newman projection, before elimination and the elimination product of the following reaction: Br CH3ONA Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY