
Interpretation:
The elements,
Concept Introduction:
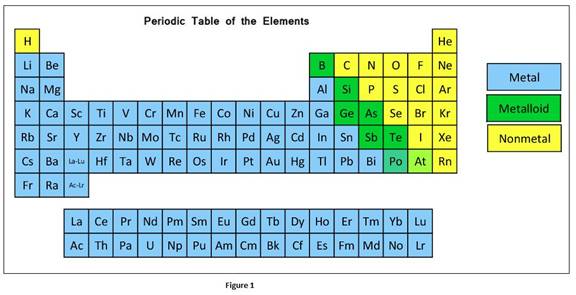
According to physical and chemical properties, the elements can be further divided into metals, non-metals and metalloids.
In a group, the metallic character of an element increases from top to bottom whereas in a period, it decreases from left to right.
Metals are elements that usually contain 1-3 outer shell electrons. Metals look shining and lustrous and are malleable and ductile. They are also good conductors of heat and electricity. They have low ionization energy and tend to lose electrons forming cations. They are most likely to form ionic compounds.
Nonmetals are elements that usually contain 4-8 outer shell electrons. They cannot conduct heat and electricity and are brittle. Also they have high
Metalloids are elements having intermediate properties of metals and nonmetals. Comparing to metals, metalloids are poor conductors but under certain conditions they can act as electrical conductors. They look shining and lustrous like metals yet they are brittle like nonmetals.
The classification elements in the periodic table as metals, nonmetals, or metalloids can be given as
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Chemistry
- When a nonmetal oxide reacts with water, it forms an oxoacid with the same oxidation number as the nonmetal. Give the name and formula of the oxide used to prepare each of these oxoacids: (a) hypochlorous acid; (b) chlorous acid; (c) chloric acid; (d) perchloric acid; (e) sulfuric acid; (f ) sulfurous acid; (g) nitric acid; (h) nitrous acid; (i) carbonic acid; ( j) phosphoric acid.arrow_forwardWhat ions are possible for the two largest stable elements in Group 4A(14)? How does each arise?arrow_forwardClassify each of the following as a metal, a nonmetal, or a metalloid: a. Mn b. I c. Te d. Sb e. Oarrow_forward
- 3. Classify each of the following elements as a noble gas, a representative element, a transition element, or an inner-transition element. Also state whether the element is paramagnetic or diamagnetic: (a) potassium; (b) phosphorous; (c) promethium; (d) platinum; (e) krypton.arrow_forwardWhich of the ions Mg2+, Sr2+, Ca2+, Be2+, Ba2+, or Ra2+ is the smallest? Which of the elements of the third period should have the highest 2nd ionization energy? S, P, Si, Al, Mg, Na Which of the halogen elements has the highest density in its standard state? Br, Cl, F, Atarrow_forwardBoron, atomic number 5, occurs naturally as two isotopes, 10B and 11B, with natural abundances of 19.9% and 80.1%, respectively.(a) In what ways do the two isotopes differ from each other? Does the electronic configuration of 10B differ from that of 11B? (b) Drawthe orbital diagram for an atom of 11B. Which electrons are the valence electrons? (c) Indicate three ways in which the 1s electrons inboron differ from its 2s electrons. (d) Elemental boron reacts with fluorine to form BF3, a gas. Write a balanced chemical equation forthe reaction of solid boron with fluorine gas. (e) ΔHf° for BF31g2 is -1135.6 kJ>mol. Calculate the standard enthalpy change in thereaction of boron with fluorine. (f) Will the mass percentage of F be the same in 10BF3 and 11BF3? If not, why is that the case?arrow_forward
- (a) Rank elements: Na, Mg, Al, and K, in increasing order of: (i) atomic size; (ii) ionization energy, and (iii) reactivity. (b) Explain why atomic size decreases from left to right, but increases from top to bottom; (c) Explain why ionization energy increases from left to right, but decreases from top to bottom; (d) Explain why the reactivity of alkali metals (Group-1) increases from top to bottom, where as the reactivity of halogen (Group-17) decreases from top to bottom.arrow_forwardThe ionization energy of an alkali metal is reflected in its reaction with water, where a bigger explosion indicates an easier reaction. In general, if X is an alkali metal, the reaction with water is: X (s) + H2O (l) = XOH (aq) + H2 (g). How does the reaction relate to the ionization of X? Write the reaction for X in the boxes below. This reaction does not need to be balanced. Any other species and the state of the reactants and products can be ignored. Include the formal charge in the superscript box. If there is no formal charge, write "0".arrow_forwardWhich of the following elements would you expect to have the largest first ionization energy? Select one: Be, Li, K, Caarrow_forward
- A hydrogen-like ion is an ion containing only one electron. The energies of the electron in a hydrogen-like ion are given by (2.180 × 10-¹5 1) 2² (1) J) E = kJ where n is the principal quantum number, and Z is the atomic number of the element. Calculate the ionization energy, in your answer to 4 significant digits. mol Note: Reference the Fundamental constants table for additional information. ·2+ of the Li²+ ion. Round Iarrow_forwardWhich of the following atoms and ions is (are) isoelectronic with Si: Ar, S2+, Ne, Al3+, P3−, As3+?arrow_forwardIs bromine (Br) a metal, a nonmetal, or a metalloid? Does bromine have a higher or lower electron affinity than selenium?arrow_forward
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





