
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 62P
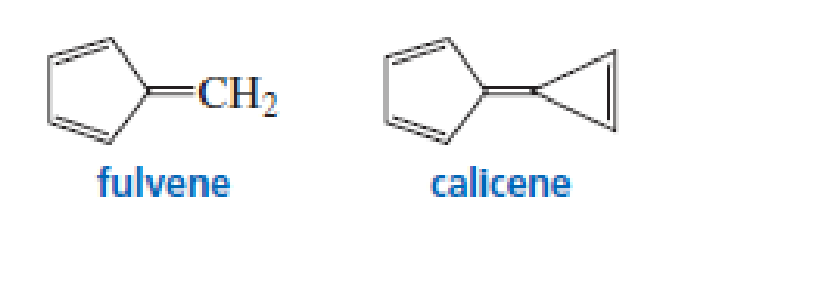
- a. In what direction is the dipole moment in fulvene? Explain.
- b. In what direction is the dipole moment in calicene? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The purine heterocycle occurs commonly in the structure of DNA.
a. How is each N atom hybridized?
b. In what type of orbital does each lone pair on a N atom reside?
c. How many a electrons does purine contain?
d. Why is purine aromatic?
purine
a.Draw all reasonable resonance structures for pyrrole, and explain why pyrrole is less resonance stabilized than benzene.
b.Draw all reasonable resonance structures for furan, and explain why furan is less resonance stabilized than pyrrole.
Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia.
a.In what type of orbital does the lone pair on each N atom in the heterocycle reside?
b. Explain why the bicyclic ring system that contains both N atoms is aromatic.
c.Draw all reasonable resonance structures for the bicyclic ring system.
Chapter 7 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 7.4 - Prob. 1PCh. 7.5 - Prob. 3PCh. 7.6 - a. Predict the relative bond lengths of the three...Ch. 7.6 - Prob. 5PCh. 7.6 - Prob. 6PCh. 7.7 - Prob. 7PCh. 7.7 - Prob. 8PCh. 7.7 - Prob. 9PCh. 7.7 - Prob. 10PCh. 7.8 - Which member of each pair is the stronger acid? a....
Ch. 7.8 - Which member of each pair is the stronger base? a....Ch. 7.9 - Prob. 13PCh. 7.9 - Which species in each of the following pairs is...Ch. 7.9 - Prob. 16PCh. 7.11 - Prob. 18PCh. 7.11 - Prob. 19PCh. 7.11 - Prob. 20PCh. 7.11 - Prob. 21PCh. 7.11 - Prob. 22PCh. 7.12 - Prob. 23PCh. 7.12 - Prob. 24PCh. 7.15 - Prob. 25PCh. 7.15 - Which of the following are aromatic? A...Ch. 7.17 - Prob. 29PCh. 7.17 - Prob. 30PCh. 7 - Prob. 31PCh. 7 - Prob. 32PCh. 7 - Prob. 33PCh. 7 - Prob. 34PCh. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - Which of the compounds in each of the following...Ch. 7 - Prob. 39PCh. 7 - Prob. 40PCh. 7 - Prob. 41PCh. 7 - Prob. 42PCh. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Prob. 45PCh. 7 - Which species in each of the pairs in Problem 45...Ch. 7 - Rank the following anions in order from most basic...Ch. 7 - a. Which oxygen atom has the greater electron...Ch. 7 - Prob. 49PCh. 7 - Which compound is the strongest base?Ch. 7 - Prob. 51PCh. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Prob. 55PCh. 7 - a. Which dienophile in each pair is more reactive...Ch. 7 - Prob. 57PCh. 7 - Draw the product of each of the following...Ch. 7 - Prob. 59PCh. 7 - Prob. 60PCh. 7 - Prob. 61PCh. 7 - a. In what direction is the dipole moment in...Ch. 7 - Propose a mechanism for each of the following...Ch. 7 - Prob. 1PCh. 7 - Prob. 2PCh. 7 - Prob. 3PCh. 7 - Prob. 4PCh. 7 - Prob. 5PCh. 7 - Prob. 6PCh. 7 - Prob. 7PCh. 7 - Prob. 8PCh. 7 - Prob. 9PCh. 7 - Prob. 10PCh. 7 - Prob. 11PCh. 7 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. Which compound would you expect to have a greater dipole moment, methyl acetate or butanone?b. Which would you expect to have a higher boiling point?arrow_forwardWhich compound is not aromatic? A. only A and D B. A, B and C C. only B D. only Aarrow_forward2. Draw the each structures. For each structure, obtain the following information:a. Chargeb. Bond dipolesc. Over-all dipolearrow_forward
- Label each compound as aromatic, antiaromatic, or not aromatic. Assume all completely conjugated rings are planar. Å a. b. C. d.arrow_forwardplease explain why A more acidic than Darrow_forwardDraw the resonance structure of the ff. and identify if aromatic or nonaromatic through Huckel’s rule. note: I need the answer immediately. I will send a good rate right away as well.arrow_forward
- Circle the molecule in each pair that is more water-soluble. 0: (c) vs. (b) VS. H VS. (a) X-H (d) -H (e) VS. (f) VS. ïarrow_forwardWhy is the center of the electrostatic potential map of benzene more red than the center of the electrostatic potential map of pyridine?arrow_forwardProline is an unusual amino acid because its N atom on the a carbon is part of a five-membered ring. a. Draw both enantiomers of proline. b. Draw proline in its zwitterionic form. -COOH prolinearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY