
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 5, Problem 14Q
Consider these three hydrocarbons:
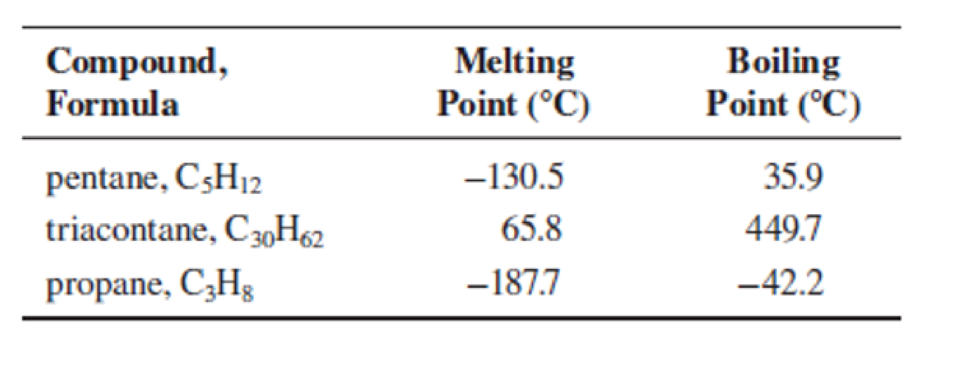
At room temperature (25°C), categorize each one as a solid, liquid, or gas.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
According to one of the principles of combustion, solid and liquid fuels must be changed to a gas before they burn. For liquid fuels, this is easily explained as liquids vaporize when enough heat is applied to reach the boiling point. How can you explain the same principle of combustion with regards to solid fuels like coal?Answer in less than 100 words.
2. Was there any physical change in the characteristics of trichloroethane when it was added to water?
As the temperature increases, which state of matter are the molecules in?
Chapter 5 Solutions
Chemistry In Context
Ch. 5.1 - Prob. 5.1YTCh. 5.1 - Prob. 5.2YTCh. 5.1 - Consult the interactive trends found in the...Ch. 5.1 - Prob. 5.4YTCh. 5.2 - For each of the fuels below, write the balanced...Ch. 5.2 - Prob. 5.6YTCh. 5.3 - Prob. 5.7YTCh. 5.4 - Prob. 5.8YTCh. 5.4 - Prob. 5.9YTCh. 5.4 - Scientific Practices Coal Versus Ethanol On the...
Ch. 5.5 - Prob. 5.12YTCh. 5.5 - Prob. 5.13YTCh. 5.6 - Although power plants require several steps to...Ch. 5.7 - Prob. 5.15YTCh. 5.7 - Prob. 5.16YTCh. 5.7 - An input of energy can be used to decrease entropy...Ch. 5.8 - Prob. 5.18YTCh. 5.8 - Prob. 5.19YTCh. 5.8 - Prob. 5.20YTCh. 5.8 - Prob. 5.21YTCh. 5.10 - Prob. 5.22YTCh. 5.11 - The combustion of one gram of natural gas releases...Ch. 5.11 - a. During the extraction of natural gas, the...Ch. 5.12 - Prob. 5.25YTCh. 5.13 - Prob. 5.26YTCh. 5.13 - Beginning in the 1920s, the octane-booster...Ch. 5.15 - Prob. 5.28YTCh. 5.15 - Prob. 5.29YTCh. 5.16 - Prob. 5.30YTCh. 5.17 - Have you ever been served cherries Jubilee or...Ch. 5.17 - Prob. 5.34YTCh. 5 - Prob. 1QCh. 5 - Prob. 2QCh. 5 - Prob. 3QCh. 5 - Energy exists in different forms in our natural...Ch. 5 - A coal-burning power plant generates electrical...Ch. 5 - Prob. 6QCh. 5 - Prob. 7QCh. 5 - Prob. 8QCh. 5 - Mercury (Hg) is present in trace amounts in coal,...Ch. 5 - Prob. 10QCh. 5 - Here are the condensed structural formulas for two...Ch. 5 - Prob. 12QCh. 5 - Prob. 13QCh. 5 - Consider these three hydrocarbons: At room...Ch. 5 - During petroleum distillation, kerosene and...Ch. 5 - Prob. 16QCh. 5 - a. Write the balanced chemical equation for the...Ch. 5 - Prob. 18QCh. 5 - Prob. 19QCh. 5 - State whether these processes are endothermic or...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Ethanol can be produced by fermentation. Another...Ch. 5 - Here are structural formulas for ethane, ethene...Ch. 5 - These three compounds all have the same chemical...Ch. 5 - Catalysts speed up cracking reactions in oil...Ch. 5 - Explain why cracking is a necessary part of the...Ch. 5 - Consider this equation representing the process of...Ch. 5 - Prob. 29QCh. 5 - Consider these three alcohols: methanol, ethanol,...Ch. 5 - Prob. 31QCh. 5 - Prob. 32QCh. 5 - Prob. 33QCh. 5 - Compare and contrast a molecule of biodiesel with...Ch. 5 - Use Figure 5.6 to compare the energy released for...Ch. 5 - Prob. 36QCh. 5 - The sustainability of burning coal (and other...Ch. 5 - In this chapter, we approximated the chemical...Ch. 5 - Prob. 39QCh. 5 - Compare the processes of combustion and...Ch. 5 - How might you explain the difference between...Ch. 5 - Write a response to this statement: Because of the...Ch. 5 - The concept of entropy and probability is used in...Ch. 5 - Bond energies such as those in Table 5.1 are...Ch. 5 - Use the bond energies in Table 5.1 to explain why...Ch. 5 - Prob. 46QCh. 5 - Prob. 47QCh. 5 - Prob. 48QCh. 5 - Prob. 49QCh. 5 - Prob. 50QCh. 5 - Prob. 51QCh. 5 - Prob. 52QCh. 5 - Prob. 53QCh. 5 - Use a diagram to show the relationship among these...Ch. 5 - On a timescale of a few years, the combustion of...Ch. 5 - Emissions of some pollutants are lower when...Ch. 5 - Although coal contains only trace amounts of...Ch. 5 - Prob. 58QCh. 5 - An article in Scientific American pointed out that...Ch. 5 - C. P. Snow, a noted scientist and author, wrote an...Ch. 5 - Chemical explosions are very exothermic reactions....Ch. 5 - Prob. 63QCh. 5 - Tetraethyllead (TEL) was first approved for use in...Ch. 5 - Tetraethyllead (TEL) has an octane rating of 270....Ch. 5 - Another type of catalyst used in the combustion of...Ch. 5 - Figure 5.8 shows energy differences for the...Ch. 5 - Prob. 68Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why is tap water a homogenous mixture, whereas distilled water is a pure substance?arrow_forwardIn a solid, any two adjacent molecules remain adjacent to one another for a a long time (minutes or longer). A molecular-scale snapshot of the position of molecules in the liquid phase would look more like a snapshot of an amorphous solid than a snapshot of a crystalline solid. At higher temperatures, a higher fraction of molecules in a liquid have enough energy to overcome the attractive forces between the molecules. In a liquid near room temperature, a given molecule spends a long time (minutes or longer) next to the same adjacent molecule before they slip away from one another. Also..! I think I know this next one already. But i Just wanted to check what you thought? If you agree? For most compounds, which is the proper order of density for highest to lowest? O liquid, solid, gas gas, liquid, solid solid, liquid, gas gas, solid, liquidarrow_forwardWhy is arrangement of the constituent atoms or molecules are important in determining the properties of a solid than a liquid or a gas?arrow_forward
- Take some water with a straw and put a few drops on plastic sheet. (a) What is the shape of the drop? (b) Move a drop around with your straw. Does the drop change? 2. Move one of the drops close to another one with your straw. What happens when two drops meet? 3. Put a small amount of one of the solids (salt, pepper, sugar, talcum powder) on one of the drops. Does the shape change? 4. Try this again with the other solids.arrow_forwardA white substance melts at 70 degrees Celsius. As a solid or in aqueous form it does not conduct electricity and can be easily crushed . What type of solid is this substance likely to be ? Justify your answer .arrow_forwardWhat type of a system and process does water heater that takes in cold water and releases out hot waterarrow_forward
- True or False. Acetone and alcohol can be used by the human body as energy sources even though in certain concentrations, they are poisonous.arrow_forwardhydrogen peroxide → water + oxygen List two physical / chemical properties for each compoundarrow_forward3. Calculate the amount of energy absorbed when 42 g of water at 21°C heats to 94°C. Did a phase change occur? If so, which one? Is this endothermic or exothermic? peararrow_forward
- Sarin is a nerve gas that was previously used as a chemical weapon. Considering the physical properties of sarin shown in the table, which statement about sarin is FALSE? Melting point Bolling point -56°C 158°C O A sample of sarin at 0°C exists as a liquid. O A sample of sarin at room temperature (22°C) exists as a liquid. O A sample of sarin at -100°C exists as a solid. O A sample of sarin at 100°C exists as a gas. O A sample of sarin at 170°C exists as a gas.arrow_forwardWhich of the following describes what occurs as a liquid becomes a solid? A physical change occurs; the particles of the liquid vibrate faster and their kinetic energy increases. A physical change occurs; the particles of the liquid vibrate more slowly and their kinetic energy decreases. A chemical change occurs; the particles of the liquid vibrate faster and their kinetic energy increases. A chemical change occurs; the particles of the liquid vibrate more slowly and their kinetic energy decreases.arrow_forwardClassify the different properties of petroleum products and explain the significance of measuring each property.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY