
Concept explainers
Consider the substituted cyclohexane shown in the ball-and-stick model.
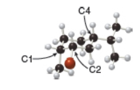
a. Label the substituents on
b. Are the substituents on
c. Are the substituents on
d. Draw the second possible conformation in the chair form, and classify it as more stable or less stable than the conformation shown in the three-dimensional model.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Basic Chemistry
General, Organic, & Biological Chemistry
Chemistry: Atoms First
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Basic Chemistry (5th Edition)
- a model of cyclohexane in a chair conformation, and explain why the names “axial” and“equatorial’ are appropriate.arrow_forwardConsider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.arrow_forwardOn the left is a stereorepresentation of glucose (we discuss the structure and chemistry of glucose in Chapter 25). (a) Convert the stereorepresentation on the left to a planar hexagon representation. (b) Convert the stereorepresentation on the left to a chair conformation. Which substituent groups in the chair conformation are equatorial? Which are axial?arrow_forward
- Label each ring in Figure 7.2 cis or trans.arrow_forwardConsider 1,2-dimethylcyclohexane.a. Draw structures for the cis and trans isomers using a hexagon for the six-membered ring.b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable?c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable?d. Which isomer, cis or trans, is more stable and why?arrow_forwardFor each compound drawn below: a. Draw representations for the cis and trans isomers using a hexagon for the six-membered ring, and wedges and dashes for substituents. b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable? c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable? d. Which isomer, cis or trans, is more stable and why? [1] [2] [3]arrow_forward
- For each compound drawn below: a. Draw representations for the cis and trans isomers using a hexagon for the six-membered ring, and wedges and dashed wedges for substituents. b.Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable? c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable? d.Which isomer, cis or trans, is more stable and why?arrow_forwardg. Draw two chair conformations of the following molecule and indicate which one is more stable. h. Draw the Newman projection of the following molecules. Classify each conformation as staggered or eclipsed around the indicated bond, and rank the conformations in order of increasing stability. CH2CH2CH3 CH3 (CH3)2CH, CH2CH2CH3 (CH3)2CH .C- CH3 CH3 CH3 Aarrow_forwardFor each compound drawn below: a.Label each OH, Br, and CH3 group as axial or equatorial. b.Classify each conformation as cis or trans.c.Translate each structure into a representation with a hexagon for the six-membered ring, and wedges and dashed wedges for groups above and below the ring. d. Draw the second possible chair conformation for each compound.arrow_forward
- a. b. For the following two pairs of molecules, (1) Draw out the chair conformation for each molecule, flip the ring if it is possible. (2) Compare both molecules to circle out which one is more stable. (3) Identify their relationship as: constitutional isomer, conformational isomer, stereoisomer or identical. (4) Find all the chiral center on each molecule and label them. Br.. Ax < and and H. H H Br H Harrow_forwarda. Draw the two chair conformers for each of the stereoisomers of trans-1-tert-butyl-3-methylcyclohexane. b. For each pair, indicate which conformer is more stable.arrow_forwardDraw the more stable chair conformation for each compound. a. trans-1-isopropyl-3-methylcyclohexane b.cis-1-sec-butyl-4-ethylcyclohexane c. cis-1-ethyl-2-isobutylcyclohexane d. trans-1,2-dibutylcyclohexanearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

