
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.8, Problem CQ
The amino acids cysteine and serine are shown. What are the Cahn-Ingold-Prelog stereochemical descriptors for these two amino acids?
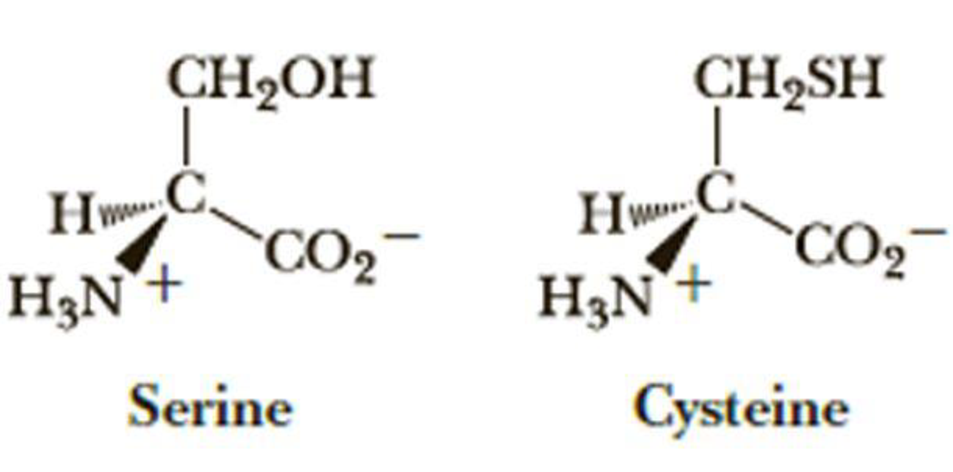
- 1. Serine is S while cysteine is R.
- 2. Cysteine is S while serine is R.
- 3. Cysteine and serine are both R.
- 4. Cysteine and serine are both S.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What is the pI of glycine? It’s structure and pKa values are shown below
what type of isomers are the compounds? which compound have a pair of enantiomers? pls explain and show the enantiomers
. Identify the following antioxidant molecule. A. alpha tocopherol hydroquinone
dehydroascorbic acid
CH3 CH3 C. alpha tocopherol semi-quinone
\ / D. ascorbic acid
C = C E. alpha tocopherol quinone
/ \
: O – C C – O CH3
\\ // \ /
C – C C – CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH3
/ \ / l l l
CH3 CH2– CH2 CH3 CH3 CH3
Chapter 3 Solutions
Organic Chemistry
Ch. 3.2 - Prob. 3.1PCh. 3.3 - Assign priorities to the groups in each set. (a)...Ch. 3.3 - Prob. 3.3PCh. 3.4 - Following are stereorepresentations for the four...Ch. 3.4 - Prob. 3.5PCh. 3.4 - Prob. 3.6PCh. 3.5 - How many stereoisomers exist for...Ch. 3.5 - How many stereoisomers exist for...Ch. 3.7 - Prob. 3.9PCh. 3.7 - Prob. 3.10P
Ch. 3.8 - If the side chain of the amino add is a methyl...Ch. 3.8 - Prob. BQCh. 3.8 - The amino acids cysteine and serine are shown....Ch. 3.8 - Prob. DQCh. 3.8 - As stated, proteins are stereochemically pure...Ch. 3.8 - As stated, proteins are stereochemically pure...Ch. 3 - Prob. 3.11PCh. 3 - One reason we can be sure that sp3-hybridized...Ch. 3 - Which compounds contain chiral centers? (a)...Ch. 3 - Prob. 3.15PCh. 3 - Prob. 3.16PCh. 3 - Prob. 3.17PCh. 3 - Mark each chiral center in the following molecules...Ch. 3 - Prob. 3.19PCh. 3 - Assign priorities to the groups in each set. (a) H...Ch. 3 - Following are structural formulas for the...Ch. 3 - Following is a staggered conformation for one of...Ch. 3 - Prob. 3.23PCh. 3 - When oxaloacetic acid and acetyl-coenzyme A...Ch. 3 - Prob. 3.25PCh. 3 - Mark each chiral center in the following molecules...Ch. 3 - Prob. 3.27PCh. 3 - Prob. 3.28PCh. 3 - Prob. 3.29PCh. 3 - Prob. 3.30PCh. 3 - Which of the following are meso compounds?Ch. 3 - Prob. 3.32PCh. 3 - Prob. 3.33PCh. 3 - Which of the following compounds are chiral?...Ch. 3 - Prob. 3.35PCh. 3 - Prob. 3.36PCh. 3 - Prob. 3.37PCh. 3 - The chiral catalyst (R)-BINAP-Ru is used to...Ch. 3 - Prob. 3.39P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is a configurational isomer?arrow_forwardb. In the human body, cysteine is oxidized to cystine, a disulfide. Draw a structural formula for cystine. O o You do not have to consider stereochemistry. o You do not have to include lone pairs in your answer. o You do not have to explicitly draw H atoms. 11... / ▼ n [F ? Ⓡ ChemDoodlearrow_forward7. Explain why 2-phenylethyl boronic acid (Compound A) is an inhibitor of chymotrypsin, which is an enzyme that catalyzes the hydrolysis of peptide linkages. What type of inhibition do you think is exemplified here? OH -CH2-CH2 B HO, Compound Aarrow_forward
- Analyze the structure of the two stereoisomers and then determine whether one or both of them is (are) chiral (s). Explain your reasoning.arrow_forwardClassify each whether it is symmetrical or unsymmetrical anhydride.arrow_forwardfind the cfse for: 1. [Rh(acac)3] 2. [PdCl2(PPh3)2] 3. [NiCl4]2-arrow_forward
- Identify the following antioxidant molecule. A. alpha tocopherol hydroquinone B.dehydroascorbic acid CH3 CH3 C. alpha tocopherol semi-quinone \ / D. ascorbic acid C = C E. alpha tocopherol quinone / \ : O – C C – O CH3 \\ // \ / C – C C – CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH3 / \ / l l l CH3 CH2– CH2…arrow_forwardAmino acids can be synthesized by reductive amination. Draw the structure of the organic compound that you would use to synthesize glutamic acid. •. You do not have to consider stereochemistry. • Draw the molecule with ionizable groups in their uncharged form. • In cases where there is more than one answer, just draw one. -85 0- ? ChemDoodle *****arrow_forward3. Draw the structure of the cross-link expected between a tyrosine side chain and the sulfonated diazonium ion shown below, CD C3 ANS H20arrow_forward
- Question 22 Based on the number of chirality centers of the molecule shown below, the maximum possible number of stereoisomers is OH NHCH; ephe drine OA. 6 OB. 2 OC.4 OD.8 ОЕ. 32 Type here to search 立arrow_forwardPlease highlight the chiral centers clearly in red, thank youarrow_forward1. Consider the following compounds. Br Br Assign stereochemistry to all of the chiral centers in both of the molecules. Do they have the same melting point? Explain your answer. 2. How could you separate a racemic mixture into two enantiopure samples?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Lanthanoids and its Position in Periodic Table - D and F Block Elements - Chemistry Class 12; Author: Ekeeda;https://www.youtube.com/watch?v=ZM04kRxm6tY;License: Standard YouTube License, CC-BY