
(a)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
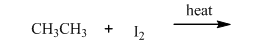
Concept introduction: Tertiary
The phenomenon of hyperconjugation refers to donation of
(b)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
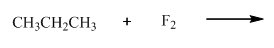
Concept introduction: Tertiary
The phenomenon of hyperconjugation refers to donation of
(c)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
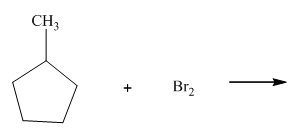
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
(d)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
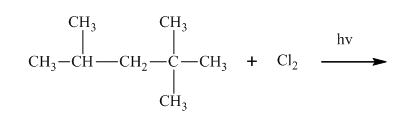
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
(e)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
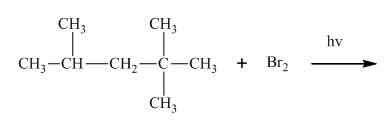
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- III) Propose a reasonable, selective synthesis of the compound below from benzene and any other reagents with four or fewer carbon atoms. If a proposed EAS reaction gives multiple regioisomers, draw them all and indicate which will be used in subsequent reactions. ZONarrow_forwardprovide the mechanism given the product and starting material along with any reagents neededarrow_forwardHelp me in drawing synthons and the synthetic equivalent for the following compounds of a,b,c and d. Thanks in advance.arrow_forward
- OH Please list the reagents needed and in what order as well as a detailed arrow-pushing mechanism showing all intermediates. Harrow_forwardProvide a reasonable mechanism for the following transformation. Includeall intermediates as well as arrows showing the making and breaking of bondsarrow_forwardWhich one of the molecules shown below prefers to exist as its enol tautomer?arrow_forward
- Draw the major organic substitution product or products for (2R,3S)-2-bromo-3-methylpentane reacting with the given nucleophile. Clearly drawn the stereochemistry, including a wedged bond, a dashed bond and two in-plane bonds at each stereogenic center. Omit any byproducts. Br CH;CH20 (conc.) H.arrow_forward(s Hq Bv write or draw the synthetic step to get to the following compounds. (Grignard reaction)arrow_forwardWhat is the best way to remeber the differences between Sn1, Sn2, E1 and E2 reations?arrow_forward
- 2, Solvolysis is referring to substitution reastions here The Solnenk i's study in ves kigaked the meehansm of solwolysis of varlous benzyl Chlorider. also the nucleophile, A diffenent, Adiffenent Hzo > SNI compound A > SN2 The Study Concluded that Compound A proceeded vin sNI pathway while eemp onnd B procelded Va sNa pathway a sUggest an explanation as to that is the case. 2 support your the SNI reaction of componnd A nexplanatiion bey gin'ng the full melhanism ofarrow_forwardTautomerization is a process in which an enol yields a ketone Give one examplefor a keto to enol tautomerization. Which form is more stable. Also, which ismore stable the E isomer or the Z isomer of 2-Chloro, 2-Butenearrow_forwardIt is often helpful to have more than one way to make a given molecule, as you may not know ahead of time which strategy will be more successful.... 4-Hexyne-2-ol can be made from propene in two different ways – one by way of a bromohydrin, using a protecting group strategy, and another by using an epoxide (which does not need a protecting group strategy). (a) First, give the synthesis which goes through the bromohydrin and uses a protecting group. OH (b) Now, modify the synthesis (you may start with propene, or any other molecule you already made in part (a)) to accomplish the synthesis of 4-hexyne-2-ol using an epoxide.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


