
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 28.37P
Convert each ball-and-stick model to a Fischer projection.
a.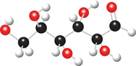 b.
b. 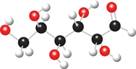
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Convert each ball-and-stick model to a Fischer projection,
a.
b.
Convert each compound to a Fischer projection formula.
Сно
но
b.
a.
HO.
HOCH, CHO
HOCH,
Convert each three-dimensional representation into a Fischer projection.
Chapter 28 Solutions
Organic Chemistry
Ch. 28 - Prob. 28.1PCh. 28 - Prob. 28.2PCh. 28 - Label each stereogenic center as R or S. a. b. c....Ch. 28 - Convert the ball-and-stick model to a Fischer...Ch. 28 - Prob. 28.5PCh. 28 - Prob. 28.6PCh. 28 - Prob. 28.7PCh. 28 - Prob. 28.8PCh. 28 - Prob. 28.9PCh. 28 - Prob. 28.10P
Ch. 28 - Prob. 28.11PCh. 28 - Prob. 28.12PCh. 28 - Prob. 28.13PCh. 28 - Prob. 28.14PCh. 28 - Prob. 28.15PCh. 28 - Prob. 28.16PCh. 28 - Prob. 28.17PCh. 28 - Draw a stepwise mechanism for the following...Ch. 28 - Prob. 28.19PCh. 28 - Prob. 28.20PCh. 28 - Prob. 28.21PCh. 28 - Prob. 28.22PCh. 28 - Draw the products formed when D-arabinose is...Ch. 28 - Prob. 28.24PCh. 28 - Prob. 28.25PCh. 28 - Prob. 28.26PCh. 28 - Prob. 28.27PCh. 28 - Prob. 28.28PCh. 28 - Prob. 28.29PCh. 28 - Prob. 28.30PCh. 28 - Prob. 28.31PCh. 28 - Prob. 28.32PCh. 28 - Prob. 28.33PCh. 28 - Prob. 28.34PCh. 28 - Problem-28.35
Draw the structures of the...Ch. 28 - Prob. 28.36PCh. 28 - 28.37 Convert each ball-and-stick model to a...Ch. 28 - Prob. 28.38PCh. 28 - Prob. 28.39PCh. 28 - 28.40 Convert each compound to a Fischer...Ch. 28 - Prob. 28.41PCh. 28 - Prob. 28.42PCh. 28 - 28.43 Draw a Haworth projection for each compound...Ch. 28 - Prob. 28.44PCh. 28 - 28.45 Draw both pyranose anomers of each...Ch. 28 - Prob. 28.46PCh. 28 - Prob. 28.47PCh. 28 - Prob. 28.48PCh. 28 - Prob. 28.49PCh. 28 - 28.50 Draw the products formed when D-altrose is...Ch. 28 - Prob. 28.51PCh. 28 - Prob. 28.52PCh. 28 - Prob. 28.53PCh. 28 - Prob. 28.54PCh. 28 - Prob. 28.55PCh. 28 - Prob. 28.56PCh. 28 - Prob. 28.57PCh. 28 - 28.58 Draw a stepwise mechanism for the following...Ch. 28 - Prob. 28.59PCh. 28 - Prob. 28.60PCh. 28 - Prob. 28.61PCh. 28 - Prob. 28.62PCh. 28 - Prob. 28.63PCh. 28 - Prob. 28.64PCh. 28 - Prob. 28.65PCh. 28 - Prob. 28.66PCh. 28 - Prob. 28.67PCh. 28 - Prob. 28.68PCh. 28 - Prob. 28.69PCh. 28 - Prob. 28.70PCh. 28 - 28.71 Draw a stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- k Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. If the reaction results in a mixture of ortho and para isomers, draw only the para-product. AICI 3 CH3C(=O)CI (1 equiv) Select to Draw Q I I I I I Problem 36 of 19 Please select a drawing or reagent from the question areaarrow_forwardDraw the eight constitutional isomers having the molecular formula C5H11Cl.a. Give the IUPAC name for each compound (ignoring R and S designations).b. Classify each alkyl halide as 1°, 2°, or 3°.c. Label any stereogenic centers.d. For each constitutional isomer that contains a stereogenic center, draw all possible stereoisomers, and label each stereogenic center as R or S.arrow_forwardAnswer each question using the ball-and-stick model of compound A.a.Give the IUPAC name for A, including R,S designations for stereogenic centers. b.Classify A as a 1°, 2°, or 3° alcohol. c.Draw a stereoisomer for A and give its IUPAC name. d.Draw a constitutional isomer that contains an OH group and give its IUPAC name. e.Draw a constitutional isomer that contains an ether and give its IUPAC name. f.Draw the products formed (including stereochemistry) when A is treated with each reagent: [1] NaH; [2] H2SO4; [3] POCl3, pyridine; [4] HCl; [5] SOCl2, pyridine; [6] TsCl, pyridinearrow_forward
- Answer each question using the ball-and-stick model of compound A. a. Give the IUPAC name for A, including R,S designations for stereogenic centers. b. Classify A as a 1°, 2°, or 3° alcohol. c. Draw a stereoisomer for A and give its IUPAC name. d. Draw a constitutional isomer that contains an OH group and give its IUPAC name. e. Draw a constitutional isomer that contains an ether and give its IUPAC name. f. Draw the products formed (including stereochemistry) when A is treated with each reagent: [1] NaH; [2] H2SO4; [3] POCI,, pyridine; [4] HCl; [5] SOCI, pyridine; [6] TSCI, pyridine. Aarrow_forwardDraw the eight constitutional isomers having the molecular formula C5H11Cl. a.Give the IUPAC name for each compound (ignoring R and S designations). b.Classify each alkyl halide as 1°, 2°, or 3°. c.Label any stereogenic centers. d.For each constitutional isomer that contains a stereogenic center, draw all possible stereoisomers, and label each stereogenic center as R or S.arrow_forwardDraw the major products of the SN1 reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, where applicable. Ignore any inorganic byproducts. H OH HCI Q Please select a drawing or reagent from the question areaarrow_forward
- Saquinavir (trade name Invirase) is a protease inhibitor, used to treat HIV (human immunodeficiency virus). a.Locate all stereogenic centers in saquinavir, and label each stereogenic center as R or S. b.Draw the enantiomer of saquinavir. c.Draw a diastereomer of saquinavir. d.Draw a constitutional isomer that contains at least one different functional group.arrow_forward1. Label the following as a: diasteriomer, enantiomer or neither (different compound) CHO CHO a. b. HO- HO- -H H- HO- H CH₂OH CHO -OH -H CH₂OH and H- H and -OH -OH CH₂OH CHO HO- -H HO- +H CH₂OH a. b.arrow_forwardConvert each Fischer projection into a three-dimensional representation with wedges and dashed bonds.arrow_forward
- Consider the ball-and-stick model of A, and label B and C as either identical to A or an enantiomer of A.arrow_forwardConvert each compound to a Fischer projection, and label each stereogenic center as R or S.arrow_forwardA. Assign each chiral center as R or S B. draw a diastereomer of ephedrine.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY