
Concept explainers
a)
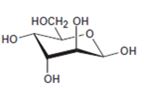
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
b)
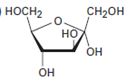
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In the α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection, while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
c)
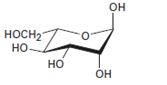
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In the α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection, while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
Trending nowThis is a popular solution!

Chapter 25 Solutions
Organic Chemistry
- Cellulose is not digestible by humans because it contains glucose units linked by -glycosidic bonds. O a-1,6 O a-1,4 O a-1,2 O B-1,4 O B-1,2arrow_forwardThe structural formula for the open-chain form of galactose is CH Н—ҫ—ОН Но—С—н НО—С—Н Н—ҫ—ОН CH-ОН (a) Is this molecule a sugar? (b) How many chiral carbons are present in the molecule? (C) Draw the structure of the six-member-ring form of this molecule.arrow_forwardCellulose, the major structural material of wood and plants, consists of a chain of B-D- glucose molecules connected by B-1,4-glycosidic bonds. In comparison, the amylose component of starch consists of a-D-glucose molecules connected by a-1,4-glycosidic bonds. Identify the following structures, denoted A and B, as the structures of cellulose or amylose. CH,OH CH2OH CH2OH CH2OH H. H H H H. H. OH H. H. H. ОН Н H Н но O. Н но H. OH H. ОН H. OH H OH CH2OH CH2OH H. CH2OH H. H. ОН Н ОН Н H CH2OH H H. ОН Н H. H OH H. H. H OH OH H H. H. ОН H. OH O B is the structure of cellulose, while A is the structure of amylose. O A and B both represent the structure of cellulose. O A and B both represent the structure of amylose. O Ais the structure of cellulose, while B is the structure of amylose.arrow_forward
- Draw the structures (using chair conformations of pyranoses) of the following disaccharides. ) 6-O-(b-d-galactopyranosyl)-d-glucopyranosearrow_forwardDraw open-chain (Fischer-projection) structure of D-(+)-glucose. What is the systematic name?arrow_forwardWhich of the following describes the following monosaccharide: CH;OH H- OH HO- CH,OH aldose and pentose ketose and pentose aldose and tetrose O ketose and tetrose O aldose and hexose O ketose and hexosearrow_forward
- C6 H 1206 is the chemical formula for a polymer of carbohydrate pentose monosaccharide hexose monosaccharide or allarrow_forwardIn addition to lactose, D-galactose and D-glucose can form different heterodisaccharides. Draw the structures (Haworth projections) of the (i) a-glucose-(1→1) -a-galactose and ii) a- glucose-(1→3)-?-galactose, two examples of heterodisaccharides that can form.arrow_forwardGlycosaminoglycans (GAGs) are heteropolysaccharides composed of repeating disaccharide units. These units have some similar characteristics that allow them to be identified as GAGS. Which of the structures are examples of glycosaminoglycans? А. C. E. HO HO COO™ H Н ОН Н CH₂OH H H H OH OH Н CH₂OH Н OH H ОН o,so H H Н CH₂OH H H ОН H CH₂OSO H NH C=0 CH, H H Н NH c=0 CH3 H CH,OH OH Н ОН OH H B. D. Н НО H ОН H CH₂OH H ОН Н СОО ОН Н Н H H OSoz H Н OH б H CH₂OH ОН CH₂OSO H ОН H Н OSO, H Н NHSO,arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





