
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25.17, Problem 33P
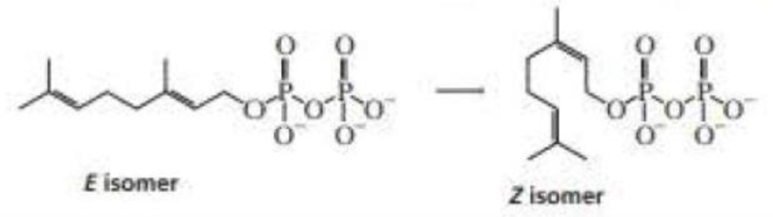
Propose a mechanism for the conversion of the E isomer of geranyl pyrophosphate to the Z isomer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which is the reductant and oxidant in the reaction?
2 KMnO4 + 5 C6H8O6 + 3 H2SO4 → 2 MnSO4 + K2SO4 + 5 C6H6O6 + 8 H2O
C6H8O6 is the reductant; KMnO4 is the oxidant
KMnO4 is the reductant; C6H8O6 is the oxidant
C6H6O6 is the reductant; MnSO4 is the oxidant
MnSO4 is the reductant; C6H6O6 is the oxidant
What is the chemicalequation of the formation ofbicarbonate from carbondioxide and water? What isthe enzyme that catalyzes thisreaction?
What happens when the codeine molecule (C18H21NO3) is exposed to air? What reactions are obtained?
Chapter 25 Solutions
Organic Chemistry
Ch. 25.6 - Prob. 2PCh. 25.6 - Prob. 3PCh. 25.6 - Why does the OH group add to the -carbon rather...Ch. 25.6 - Prob. 5PCh. 25.6 - How many molecules of NADH are formed from the...Ch. 25.7 - Prob. 7PCh. 25.7 - Prob. 8PCh. 25.7 - The oxidation of glyceraldehyde-3-phosphate to...Ch. 25.7 - Prob. 10PCh. 25.8 - Prob. 11P
Ch. 25.8 - Prob. 12PCh. 25.8 - Prob. 13PCh. 25.8 - Propose a mechanism for the reduction of...Ch. 25.9 - Prob. 15PCh. 25.9 - Prob. 16PCh. 25.10 - Acid-catalyzed dehydration reactions are normally...Ch. 25.10 - Prob. 18PCh. 25.10 - Prob. 19PCh. 25.10 - Acid-catalyzed dehydration reactions are normally...Ch. 25.10 - Prob. 21PCh. 25.10 - Prob. 22PCh. 25.11 - Prob. 23PCh. 25.12 - a. What is the name of the enzyme that converts...Ch. 25.15 - Prob. 25PCh. 25.16 - Prob. 26PCh. 25.16 - Prob. 27PCh. 25.17 - Propose mechanisms for the Claisen condensation...Ch. 25.17 - Prob. 29PCh. 25.17 - Propose a mechanism for the conversion of...Ch. 25.17 - Propose a mechanism for the biosynthesis of...Ch. 25.17 - Propose a mechanism for the conversion of the E...Ch. 25.17 - The fluoro-substitued geranyl pyrophosphate shown...Ch. 25.17 - Prob. 35PCh. 25.18 - Draw the individual 1,2-hydride and 1,2-methyl...Ch. 25 - Prob. 38PCh. 25 - Prob. 39PCh. 25 - Prob. 40PCh. 25 - Prob. 41PCh. 25 - Prob. 42PCh. 25 - Prob. 43PCh. 25 - Prob. 44PCh. 25 - Prob. 45PCh. 25 - Prob. 46PCh. 25 - Prob. 47PCh. 25 - Prob. 48PCh. 25 - Prob. 49PCh. 25 - Prob. 50PCh. 25 - Prob. 51PCh. 25 - Prob. 52PCh. 25 - Prob. 53PCh. 25 - Prob. 54PCh. 25 - Prob. 55PCh. 25 - Prob. 56PCh. 25 - Prob. 57PCh. 25 - Prob. 58PCh. 25 - Prob. 59PCh. 25 - Prob. 60PCh. 25 - Prob. 61PCh. 25 - Prob. 62PCh. 25 - UDP-galactose-4-epimerase converts UDP-galactose...Ch. 25 - A student is trying to determine the mechanism for...Ch. 25 - What would be the results of the experiment in...Ch. 25 - Prob. 66PCh. 25 - Propose a mechanism for the biosynthesis of...Ch. 25 - Eudesmol is a sesquiterpene found in eucalyptus....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The vitamin Niacin is used to form nicotinamide adenosine dinucleotide, which readily shuttles between its oxidized (NAD+) and reduced (NADH) forms. The latter serves as a cellular equivalent to NaBH4. The essential portions of the structures are shown below. Outline a mechanism for the cellular conversion of pyruvate to lactate. (Note: like NaBH4, NADH cannot reduce carboxylic acid carbonyls).arrow_forwardOne of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of sedoheptulose 7-phosphate with glyceraldehydes 3-phosphate in the presence of a transaldolase to yield erythrose 4-phosphate and fructose 6-phosphate. (a) The first part of the reaction is the formation of a protonated Schiff base of sedoheptulose 7-phosphate with a lysine residue in the enzyme followed by a retro-aldol cleavage to give an enamine plus erythrose 4-phosphate. Show the structure of the enamine and the mechanism by which it is formed. (b) The second part of the reaction is a nucleophilic addition of the enamine to glyceraldehyde 3-phosphate followed by hydrolysis of the Schiff base to give fructose 6-phosphate. Show the mechanism.arrow_forwardFor the following reaction scheme, identify by drawing the reagents a, c, and d and the intermediate b that are formed in the synthesis of 2-phenylethanoic acid.arrow_forward
- Which Compound Is Oxidized To Benzoic Acid With K2Cr2O7 In Acidic Medium?arrow_forwardThe hydrolysis of pyrophosphate to orthophosphate drives biosynthetic reactions such as DNA synthesis. In Escherichia coli, a pyrophosphatase catalyzes this hydrolytic reaction. The pyrophosphatase has a mass of 120 kDa and consists of six identical subunits. A unit of activity for this enzyme, U, is the amount of enzyme that hydrolyzes 10 umol of pyrophosphate in 15 minutes. The purified enzyme has a Vnax of 2800 U per milligram of enzyme. When (S] >> KM, how many micromoles of substrate can 1 mg of enzyme hydrolyze per second? Vnax = umol -s. mg- If cach enzyme subunit has one active site, how many micromoles of active sites, or (E]r, are there in 1 mg of enzyme? (Er = umol - mg-arrow_forwardFor the following reaction scheme, identify by drawing the reagents b, c, and e, and the intermediate d that are formed in the synthesis of the following carboxylic acid.arrow_forward
- One of the steps in the biosynthesis of uridine monophosphate is the reaction of aspartate with carbamoyl phosphate to give carbamoyl aspartate followed by cyclization to form dihydroorotate. Propose mechanisms for both steps.arrow_forwardThe reaction for the magnesium cation with 8-hydroxyquinoline is carried out in the presence of: A) Nitric acid B) Sodium hydroxide C) A solution of ammonia and ammonium chloride D) Sulfuric acid E) Sodium acetatearrow_forwardAmino acids are metabolized by a transamination reaction in which the amino group of the amino acid changes places with the keto group of an a-keto acid; the amino acid and a new a-keto acid. Draw the products of the transamination reaction between threonine and 2-oxopentanedioate. • You do not have to consider stereochemistry. • Use the charge tools to adjust the charges of the amine and carboxyl groups to the form in which they would be found at physiological pH. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu. ? √n [ Ⓡ ChemDoodlearrow_forward
- The thiamine pyrophosphate (TPP)-dependent enzyme benzoylformate decarboxylase catalyzes the decarboxylation of benzoylformate to yield benzaldehyde. Propose a mechanism based on your knowledge of TPP-dependent mechanisms. You may begin the reaction with the ylid form of TPP and you only need to show the structure of the “business end” of TPP. Show all electron flow using the arrow convention.arrow_forwardThis is a reaction scheme for epoxidation of cholesterol with MCPBA. The reaction mixture was put through the alumina in a burette to purify the epoxide product. Explain how the process works.arrow_forwardState various conditions required in preparation of H2SO4?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License