
Concept explainers
(a)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and
Answer to Problem 25.39P
The structure that corresponds to the given name is,
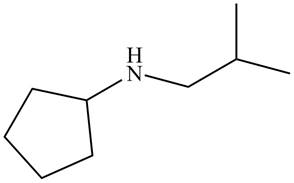
Figure 1
Explanation of Solution
The name of the given molecule suggests that a nitrogen atom is bonded to cyclopentane and isobutyl group. Cyclopentane consists of five carbon atoms. Therefore, the structure of the given compound is,
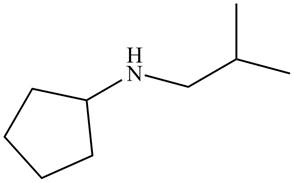
Figure 1
The structure that corresponds to the given name is shown in Figure 1.
(b)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
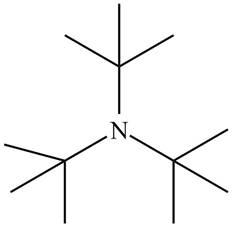
Figure 2
Explanation of Solution
The name of the given compound is tri-tert-butylamine. Prefix ‘tri’ shows that a nitrogen atom is bonded to three tert-butyl groups. Therefore, the structure of the given compound is,
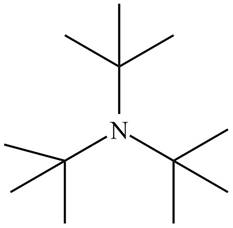
Figure 2
The structure that corresponds to the given name is shown in Figure 2.
(c)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
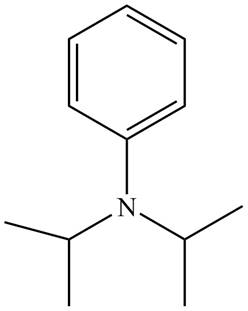
Figure 3
Explanation of Solution
The name of the given compound is N, N-diisopropylaniline. Prefix ‘di’ shows that a nitrogen atom is bonded to two isopropyl groups and it is bonded to six membered
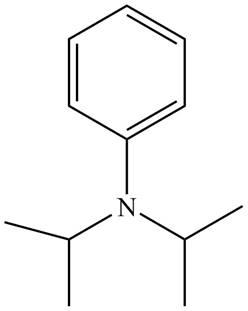
Figure 3
The structure that corresponds to the given name is shown in Figure 3.
(d)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
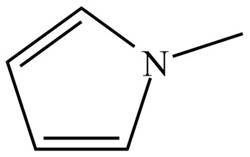
Figure 4
Explanation of Solution
The name of the given compound is N-methylpyrrole. Pyrrole is the parent chain of the given compound. It is an aromatic five membered ring which contains nitrogen atom and two double bonds. The nitrogen atom is bonded to methyl group. Therefore, the structure of the given compound is,

Figure 4
The structure that corresponds to the given name is shown in Figure 4.
(e)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
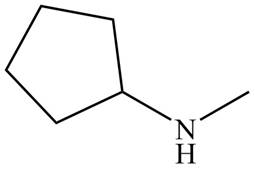
Figure 5
Explanation of Solution
The name of the given compound is N-methylcyclopentanamine. The name suggests that nitrogen atom is bonded to cyclopentane and methyl group. Therefore, the structure of the given compound is,

Figure 5
The structure that corresponds to the given name is shown in Figure 5.
(f)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
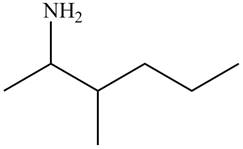
Figure 6
Explanation of Solution
The name of the given compound is

Figure 6
The structure that corresponds to the given name is shown in Figure 6.
(g)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
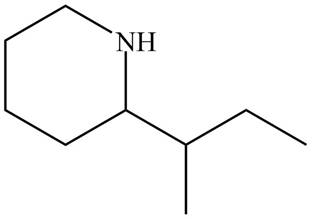
Figure 7
Explanation of Solution
The name of the given compound is

Figure 7
The structure that corresponds to the given name is shown in Figure 7.
(h)
Interpretation: The structure that corresponds to the given name is to be drawn.
Concept introduction: One should follow the given three steps to derive the structure of the compound from its IUPAC name. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 25.39P
The structure that corresponds to the given name is,
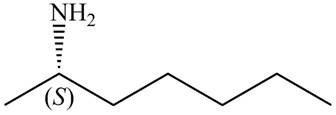
Figure 8
Explanation of Solution
The name of the given compound is (S)-heptan-

Figure 8
The structure that corresponds to the given name is shown in Figure 8.
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry
- Reduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary amine D. a secondary amine E. a primary aminearrow_forward1. What is the product of the reaction? 1) LAH 2) Hо* но OH а. b. OH с. HO. HO. d. 2. Which amine is expected to be least soluble in HzO? `NH2 а. c. N. b. d. ZIarrow_forwardWhat are the functional groups present in this antibacterial antibiotic? A. Nitro, phenyl, amine, carbonyl, hydroxyl B. Nitro, phenyl, amine, carbonyl, C. Nitro, phenyl, amine, hydroxyl D. Nitro, phenol, amine, carbonyl, hydroxyl A brief explanation would be highly appreciated + upvotearrow_forward
- Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forwardGive the structure corresponding to each name.a. cyclohexyl propanoateb. cyclohexanecarboxamidec. benzoic propanoic anhydrided. 3-methylhexanoyl chloride e. octyl butanoatef. N,N-dibenzylformamidearrow_forwardAmines will not react with which of the follow ing? alkyl halides b. carboxylic acids 52. а. с. Ketones d. hydrogen chloride Which statement below is incorrect? the smallest aldehyde has the formula HCOH a carbonyl carbon cons ists of a carbon-oxygen double bond in an aldehyde, the carbonyl carbon is always bonded to a hydrogen atom d. 53. а. b. с. Ketones are more soluble in water than alcohols with the equiva lent number of carbonsarrow_forward
- explain the solubility behavior of each representatives amine in water a. aniline b. diethylamine c. N,N-diethylanilinearrow_forward1. Complete each of the following by supplying the missing reagents. Draw thestructures of each of the reactants and products.a. N-methylpropanamide + ? → methanamine + ?b. Formamide + strong acid → ? + ? 2. Write the structure of the following: N-Propylhexanamide Propionamide N-Methylpropanamidearrow_forward83. Aliphatic amides 1. are prepared by heating the corresponding aliphatic armmonium carboxylate salt 2. will lost a-CO- group when treated wilth bromate(i) in an alkali 3. can be coverled into the corresponding acid by reacting with NANO HCI. 84. Uren (HN),CO) 1. is an amide because it contains the -C-NH, group 2. has a structure that contains the peptide link 3. reacts with NANO /HCI to give methanoic acidarrow_forward
- Draw the structure corresponding to each name. a. 2-hydroxyheptanoic acid b. 4-chlorononanoic acid c. 3,4-dibromobenzoic acid d. lithium propanoate e. 2,2-dibromobutanoic acid f. ethyl 2-methylpropanoatearrow_forwardComplete each of the following by supplying the missing reagents. Draw the structures of each of the reactants and products. a. N-Methylpropanamide + ? --> propanoic acid + ? b. N,N-Dimethylacetamide + strong acid --> ? + ? c. Formamide + strong acid --> ? + ?arrow_forwardDraw the structure corresponding to each name. a. 3,3-dimethylpentanoic acid b. 4-chloro-3-phenylheptanoic acid c. (2R)-2-chloropropanoic acid d. B,B-dichloropropionic acid e. m-hydroxybenzoic acid f. o-chlorobenzoic acid i. 2,2-dichloropentanedioic acid j. 4-isopropyl-2-methyloctanedioic acid g. potassium acetate h. sodium a-bromobutyratearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





