
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 23.5P
Which
a. 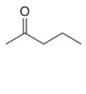 b.
b. 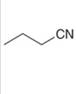 c.
c. 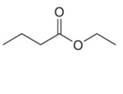 d.
d. 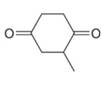
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What is the conjugate acid of C6H₂O¯ ?
A.
B.
C6H402-
C₂H4O
C. CH₂OH
ODCH₂OH
O E. C6H5OH3+
Which compound is considered as the conjugate base? *
D. OH-
B. C2H5NH+3
C. H2O
A. C2H5NH2
What is the conjugate base to H2PO−3H2PO3− ?
a. HPO2−3HPO32−
b. HPO−3HPO3−
c. PO3−3PO33−
d. H2PO−4H2PO4−
e. H3PO4H3PO4
Chapter 23 Solutions
Organic Chemistry
Ch. 23 - Problem 23.1 Draw the enol or keto tautomer(s) of...Ch. 23 - Prob. 23.2PCh. 23 - Problem 23.3 When phenylacetaldehyde is dissolved...Ch. 23 - Prob. 23.4PCh. 23 - Problem 23.5 Which bonds in the following...Ch. 23 - Prob. 23.6PCh. 23 - Prob. 23.7PCh. 23 - Prob. 23.8PCh. 23 - Prob. 23.9PCh. 23 - Prob. 23.10P
Ch. 23 - Problem 23.11 Draw the products of each...Ch. 23 - Problem 23.12 Draw the products of each reaction....Ch. 23 - Prob. 23.13PCh. 23 - Prob. 23.14PCh. 23 - Prob. 23.15PCh. 23 - Prob. 23.16PCh. 23 - Prob. 23.17PCh. 23 - Problem 23.18 How can pentan-2-one be converted...Ch. 23 - Problem 23.19 Identify A, B, and C, intermediates...Ch. 23 - Problem 23.20 Which of the following compounds...Ch. 23 - Problem 23.21 Draw the products of each...Ch. 23 - Prob. 23.22PCh. 23 - Prob. 23.23PCh. 23 - Prob. 23.24PCh. 23 - Prob. 23.25PCh. 23 - Prob. 23.26PCh. 23 - Prob. 23.27PCh. 23 - Prob. 23.28PCh. 23 - 23.29 Draw enol tautomer(s) for each compound....Ch. 23 - 22.30 The cis ketone A is isomerized to a trans...Ch. 23 - 23.31 Draw enol tautomer(s) for each compound.
...Ch. 23 - Prob. 23.32PCh. 23 - Prob. 23.33PCh. 23 - Prob. 23.34PCh. 23 - 23.35 Rank the labeled protons in each compound in...Ch. 23 - Prob. 23.36PCh. 23 - Prob. 23.37PCh. 23 - 23.38 Acyclovir is an effective antiviral agent...Ch. 23 - 23.39 Explain why forms two different alkylation...Ch. 23 - Prob. 23.40PCh. 23 - 23.41 Acid-catalyzed bromination of pentanone ...Ch. 23 - 23.42 Draw a stepwise mechanism for the following...Ch. 23 - Prob. 23.43PCh. 23 - Prob. 23.44PCh. 23 - 23.45 Devise a synthesis of valproic acid , a...Ch. 23 - 23.46 Synthesize each compound from diethyl...Ch. 23 - Prob. 23.47PCh. 23 - Prob. 23.48PCh. 23 - Prob. 23.49PCh. 23 - 23.50 Draw the organic products formed in each...Ch. 23 - 23.51 Draw the products formed (including...Ch. 23 - Prob. 23.52PCh. 23 - Prob. 23.53PCh. 23 - 23.54 Clopidogrel is the generic name for Plavix,...Ch. 23 - 23.55 What reaction conditions—base, solvent, and...Ch. 23 - Prob. 23.56PCh. 23 - 23.57 Draw a stepwise mechanism showing how two...Ch. 23 - 23.58 Draw a stepwise mechanism for the following...Ch. 23 - Prob. 23.59PCh. 23 - 23.60 Draw stepwise mechanisms illustrating how...Ch. 23 - Prob. 23.61PCh. 23 - Prob. 23.62PCh. 23 - 23.63 Synthesize each compound from cyclohexanone...Ch. 23 - Prob. 23.64PCh. 23 - Prob. 23.65PCh. 23 - 23.66 Synthesize (Z)-hept-5-en-2-one from ethyl...Ch. 23 - Prob. 23.67PCh. 23 - 23.68 Capsaicin, the spicy component of hot...Ch. 23 - 23.69 Treatment of W with , followed by , affords...Ch. 23 - Prob. 23.70PCh. 23 - Prob. 23.71PCh. 23 - Prob. 23.72PCh. 23 - Prob. 23.73PCh. 23 - Prob. 23.74P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Several acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forward1. A. The conjugate base of HCO3− is CO32− HCO3− H2CO3 B. The conjugate acid of HCO3- is CO32− HCO3− H2CO3 C. The conjugate base of H2O is OH− H2O H3O+ D. The conjugate acid of H2O is OH− H2O H3O+ E. What is the conjugated base of each of the following acids in water: HClO4; H2S; HCO3-; H2PO-4F. What is the conjugated acid of each of the following bases in water: SO4-2; H2O; HCO3-; HPO4-2arrow_forwardFor each pair of molecules or ions, select the stronger base and write its Lewis structure. Q. CH3CH2O-or H-arrow_forward
- 24. Which of the following is least basic? a. l b. II c. III d. IV e. V ملاتھ NH I - III IV ماarrow_forwardFor each pair of molecules or ions, select the stronger base and write its Lewis structure. Q.CH3NH- or CH3O-arrow_forwardWhich of the following compounds is expected to be the MOST acidic? a. H,C=CH, b. CH,NH, c. CH,CH, d. HC=CHarrow_forward
- Predict the conjugate acid of CH₃O⁻. A. CH3OH B. CH3OH2+ C. CH2O2- D. CH3Oarrow_forwarddentify the conjugate base in the reaction of propanoic acid CH3CH3COOH reacting with methyl amine, CH3NH2. a. CH3NH2 b. CH3CH3COOH c. CH3NH3+ d. OH-1 e. CH3CH3COO-1arrow_forwardFor each pair of molecules or ions, select the stronger base and write its Lewis structure. Q. NH3 or H2Oarrow_forward
- Indicate the conjugate acid of CH3NHCH3. a. CH3NNCH3 b. CH3N-CH3 c. CH3N+H2CH3 d. CH3N+HCH3 Which of the following is true about the molecule/s and acid strength ? a. HCl is a weaker than HI acid because Cl- ion is more stable than I- ion. b. H2SO4 is a strong acid because it has 2 Hydrogens c. HOCl is weaker than HOBr because Cl is more electronegative than Br d. CH3CH2OH is a weaker acid than CH3COOH because the negative charge is more dispersed in the conjugate base of CH3COOH. Given : ?? = [?3?+][??−] = 1.0 × 10−14 at 25C . pKw= 14 Which of the following is true ? a. At 25 C, [?3?+] & [??−] would be large in a water solution. b. At 25 C, the pH of water is =14 c. The pH of an aqueous solution can be determined if [OH-] is provided. d. At 25 C, majority of H2O will be dissociated. pH of 0.125 M NaOH is a. 0.903 b. 13.3 c. 13.9 d. 13.10arrow_forwardWhat is the conjugate acid of HCO3 ? O A. HCO32- O B. H2CO3 O C. CO3- O D. CO32- O E. HCO3+arrow_forwardAmong the sets of acids, which is/are correctly arranged according to INCREASING acidity? One or more sets may be chosen. Explain why. a. HBr < H2Se < AsH3 b. CH4 < PH3 < H2S c. H2S < H2Te < H2Se d. HSO3– < H2SO3 < H2SO4arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY