
Concept explainers
Interpretation:
To illustrate the stabilization of a carboxylate anion, the resonance structure and resonance hybrid structure of the carboxylate ion are to be drawn.
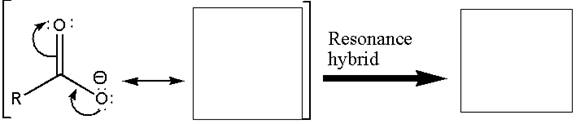
Concept introduction:
Resonance exists in species for which there are two or more valid Lewis structures. Resonance structures differ only in the placement of their valence electrons, not their atoms. Resonance structures are imaginary; the one, true species is represented by the resonance hybrid. A resonance hybrid is a weighted average of all resonance structures. Resonance provides stabilization. The resonance hybrid looks most like the most stable resonance structure. Resonance stabilization is usually large when resonance structures are equivalent.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Indicate which of the following compounds or intermediates have an aromatic character. Please provide only typed answer solution no handwritten solution needed allowedarrow_forwardPlease draw the structure CLEARLY, thanksarrow_forwardUsing the reaction shown in Box (the attack of an amine on the carbonyl group of a ketone) as a starting point, draw curved arrows to represent the base-catalyzed reaction (when the group —B: is present).arrow_forward
- Explain, which acid is less polar to most polar and arranged them in less polar?arrow_forwardIn electrophilic aromatic substitution reaction by drawing the structures of the compounds given below Expla in which products will be formed as a result of its reaction with the nitronium ion. a)methoxybenzene, b)benzoic acidarrow_forwardGive a clear explanation handwritten answer..complete the following reactionarrow_forward
- Identify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.arrow_forwardComplete the following reaction. Thank you!arrow_forwardComplete the curved arrow notation and include the structure of the missing organic intermediate for the following steps of a nucleophilic aromatic substitution via an addition–elimination mechanism.arrow_forward
- Rank the molecules in decreasing (strongest to weakest) acidity. Include ALL resonance structures with arrows and explain reasoning for rank.arrow_forwardWeak base can react with strong acid to form a salt. If pyridine reacts with a strong acid to form pyridinium salt, will the pyridinium salt be more aromatic than pyridine? .. If yes explain carefully.. If no, contrast..arrow_forwardBased on the following groups Acid chloride, Amide, Ester a) Select the most reactive group towards nucleophilic acyl substitution reaction. b) Describe the reasons in question (a). c) Identify the group(s) that can been synthesized from acid chloride.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
