
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.61P
Following are the steps in one of the several published syntheses of frontalin, a pheromone of the western pine beetle.
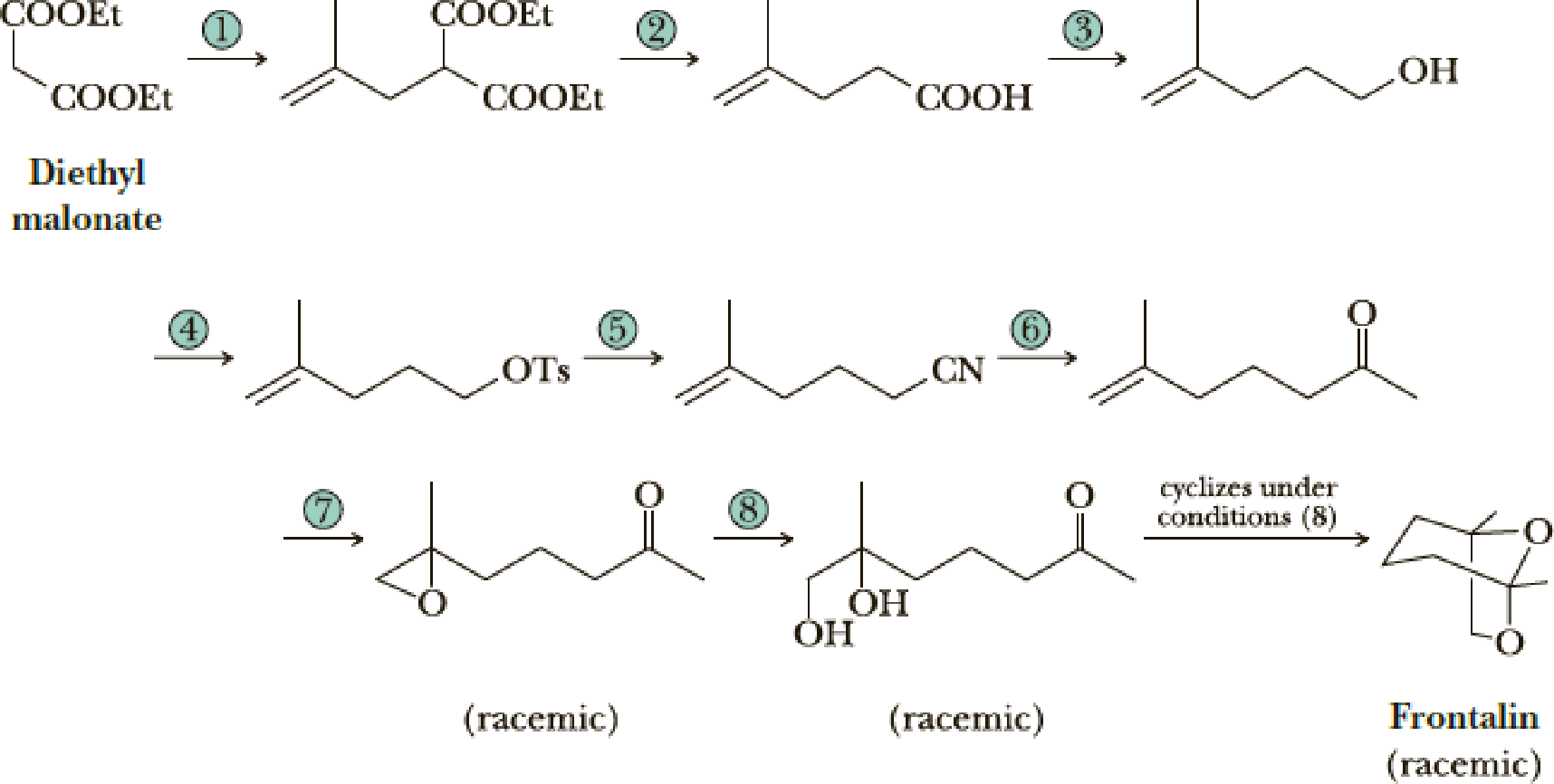
- (a) Propose reagents for Steps 1–8.
- (b) Propose a mechanism for the cyclization of the ketodiol from Step 8 to frontalin.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Propose a mechanism for the reaction of(a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane.(b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane.
The following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxide
In an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?
Chapter 19 Solutions
Organic Chemistry
Ch. 19.2 - Prob. 19.1PCh. 19.2 - Prob. 19.2PCh. 19.2 - Prob. 19.3PCh. 19.3 - Prob. 19.4PCh. 19.3 - Prob. 19.5PCh. 19.3 - Prob. 19.6PCh. 19.5 - Prob. 19.7PCh. 19.5 - Prob. 19.8PCh. 19.5 - Prob. 19.9PCh. 19.6 - Prob. 19.10P
Ch. 19.6 - Prob. 19.11PCh. 19.7 - Prob. 19.12PCh. 19.8 - Prob. 19.13PCh. 19.8 - Prob. 19.14PCh. 19.8 - Prob. 19.15PCh. 19.8 - Prob. 19.16PCh. 19.9 - Prob. 19.17PCh. 19.9 - Prob. AQCh. 19.9 - Prob. BQCh. 19.9 - Prob. CQCh. 19.9 - Prob. DQCh. 19.9 - Prob. EQCh. 19.9 - Prob. FQCh. 19.9 - Prob. GQCh. 19.9 - Intermediate G in Synthesis III is produced as a...Ch. 19.9 - Prob. IQCh. 19.9 - Prob. JQCh. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - Prob. 19.20PCh. 19 - Prob. 19.21PCh. 19 - Prob. 19.22PCh. 19 - Prob. 19.23PCh. 19 - Cyclohexene can be converted to...Ch. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - Prob. 19.28PCh. 19 - Prob. 19.29PCh. 19 - Prob. 19.30PCh. 19 - Draw structural formulas for the -ketoesters...Ch. 19 - Prob. 19.32PCh. 19 - Prob. 19.33PCh. 19 - Propose a synthesis for each ketone, using as one...Ch. 19 - Prob. 19.35PCh. 19 - Claisen condensation between diethyl phthalate and...Ch. 19 - Prob. 19.37PCh. 19 - Prob. 19.38PCh. 19 - Prob. 19.39PCh. 19 - Enamines normally react with methyl iodide to give...Ch. 19 - Prob. 19.41PCh. 19 - Prob. 19.42PCh. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49PCh. 19 - Prob. 19.50PCh. 19 - Prob. 19.51PCh. 19 - Prob. 19.52PCh. 19 - Show experimental conditions by which to carry out...Ch. 19 - Prob. 19.55PCh. 19 - The compound 3,5,5-trimethyl-2-cyclohexenone can...Ch. 19 - Prob. 19.57PCh. 19 - Prob. 19.58PCh. 19 - The widely used anticoagulant warfarin (see...Ch. 19 - Following is a retrosynthetic analysis for an...Ch. 19 - Following are the steps in one of the several...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.65PCh. 19 - Prob. 19.67PCh. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - In Problem 7.28, we saw this two-step sequence in...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Prob. 19.79PCh. 19 - Prob. 19.80PCh. 19 - Prob. 19.81PCh. 19 - The following molecule undergoes an intramolecular...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardTestosterone is one of the most important male steroid hormones. When testosterone is hydrated by treatment with acid, rearrangement occurs to yield the product shown. Propose a mechanism to account for this reaction.arrow_forward
- Reaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0 °C, followed by treatment with N,N-diethylaniline.arrow_forwardBisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction.arrow_forwardPhenylacetone can form two different enols.(a) Show the structures of these enols.(b) Predict which enol will be present in the larger concentration at equilibrium.(c) Propose mechanisms for the formation of the two enols in acid and in basearrow_forward
- 4 Predict the products of reactions of ethers and epoxides, including the following:(a) Cleavage and autoxidation of ethers(b) Acid- and base-promoted opening of epoxides(c) Reactions of epoxides with organometallic reagents(d) Cleavage of silyl ethersarrow_forwardCompound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forwardThe mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, exceptthat the nitrile is first protonated, activating it toward attack by a weak nucleophile (water).Under acidic conditions, the proton transfer (tautomerism) involves protonation on nitrogen followed by deprotonation on oxygen. Propose a mechanism for the acid-catalyzedhydrolysis of benzonitrile to benzamide.arrow_forward
- Complete the statements by providing the reagents necessary to complete each step of the synthesis. Ketone reacted with CO₂H and then yielded a derivative, which was treated with 1) (i) C6H5MgBr (ii) H3O+ 2) (i) DIBAL-H (ii). H3O+ 3) Sia2BH,H202, NaOH 4) CH2N2 (diazamethane) CO₂C2H5 5) PCC (an oxidant) 6) H2SO4, H2O, heat 7) HgSO4/ H2SO4 8) C₂H5OH, H3O*, heat TABLE OF REAGENTS to form an unsaturated carboxylic acid, treatment with to yield unsaturated aldehyde as the final product. CHO MacBook Air 9) C6H5 CH2 P(C6H5)3; NaOH 10) PCC (an oxidant) 11) NaCN, H₂SO4 12) PhCH2CHO, H3O*arrow_forwardIdentify products A and B from the given 1H NMR data. Treatment of CH2=CHCOCH3 with one equivalent of HCl forms compound A. A exhibits the following absorptions in its 1H NMR spectrum: 2.2 (singlet, 3H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2 H) ppm. What is the structure of A?arrow_forwardgas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License