
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN: 9781133104261
Author: Raymond A. Serway, John W. Jewett
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 2P
Consider Joule’s apparatus described in Figure P17.2. The mass of each of the two blocks is 1.50 kg, and the insulated tank is filled with 200 g of water. What is the increase in the water’s temperature after the blocks fall through a distance of 3.00 m?
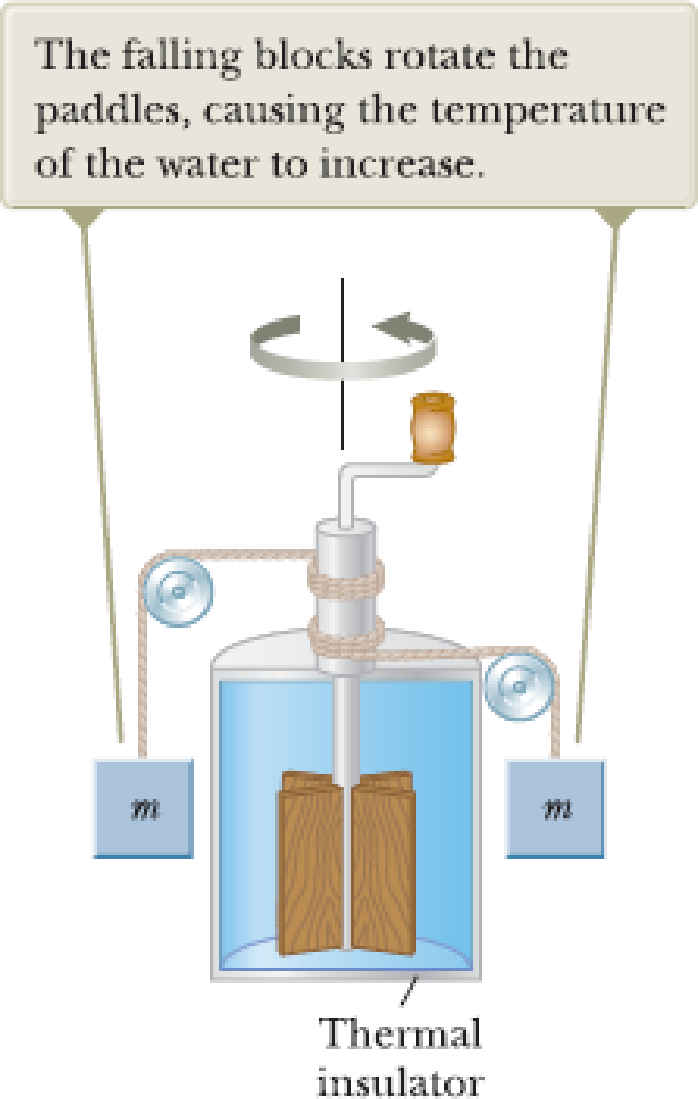
Figure P17.2.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Researchers are conducting a study to quantity the thermal conductivity of a composite material. A square box is made from 1932 cm2 sheets of the composite insulating material that is 5.2 cm thick. A 125 W heater is placed inside the box. Sensors attached to the box show that the interior and exterior surfaces of one face have reached the constant temperatures of 71°C and 23°C. What is the thermal conductivity in W/m-K?
The average thermal conductivity of the walls (including windows) and roof of a house in Figure P11.46 is 4.8 x 10-4 kW/m .°C, and their average thickness is 21.0 cm. The house is heated with natural gas, with a heat of combustion (energy released per cubic meter of gas burned) of 9300 kcal/m3. How many cubic meters of gas must be burned each day to maintain an inside temperature of 25.0°C if the outside temperature is 0.0°C? Disregard surface air layers, radiation, and energy loss by heat through the ground.
A150 cm long copper rod is 3kg in mass. What is the change in length (in centimeters) in the rod if 45000 J of energy is transferred to it? The coefficient of linear expansion of copper is 17x10^-6 /C°. The specific heat capacity of copper is 390 J/kg.C°.
Round-off your answer to the nearest thousandths. Do not type the unit of your final answer.
Chapter 17 Solutions
Principles of Physics: A Calculus-Based Text
Ch. 17.2 - Prob. 17.1QQCh. 17.3 - Prob. 17.2QQCh. 17.3 - Prob. 17.3QQCh. 17.5 - Prob. 17.4QQCh. 17.6 - Characterize the paths in Figure 17.10 as...Ch. 17.7 - (i) How does the internal energy of an ideal gas...Ch. 17.10 - Prob. 17.7QQCh. 17 - Prob. 1OQCh. 17 - A 100-g piece of copper, initially at 95.0C, is...Ch. 17 - Prob. 3OQ
Ch. 17 - Prob. 4OQCh. 17 - Prob. 5OQCh. 17 - Prob. 6OQCh. 17 - Prob. 7OQCh. 17 - Prob. 8OQCh. 17 - Prob. 9OQCh. 17 - Prob. 10OQCh. 17 - Star A has twice the radius and twice the absolute...Ch. 17 - If a gas is compressed isothermally, which of the...Ch. 17 - When a gas undergoes an adiabatic expansion, which...Ch. 17 - Ethyl alcohol has about one-half the specific heat...Ch. 17 - Prob. 15OQCh. 17 - Prob. 1CQCh. 17 - Prob. 2CQCh. 17 - Pioneers stored fruits and vegetables in...Ch. 17 - Why is a person able to remove a piece of dry...Ch. 17 - Prob. 5CQCh. 17 - Prob. 6CQCh. 17 - It is the morning of a day that will become hot....Ch. 17 - You need to pick up a very hot cooking pot in your...Ch. 17 - Rub the palm of your hand on a metal surface for...Ch. 17 - Prob. 10CQCh. 17 - Prob. 11CQCh. 17 - Prob. 12CQCh. 17 - On his honeymoon, James Joule traveled from...Ch. 17 - Consider Joules apparatus described in Figure...Ch. 17 - Prob. 3PCh. 17 - Prob. 4PCh. 17 - Prob. 5PCh. 17 - Prob. 6PCh. 17 - Prob. 7PCh. 17 - Prob. 8PCh. 17 - Prob. 9PCh. 17 - Prob. 10PCh. 17 - Prob. 11PCh. 17 - Prob. 12PCh. 17 - Prob. 13PCh. 17 - Prob. 14PCh. 17 - In an insulated vessel, 250 g of ice at 0C is...Ch. 17 - Prob. 16PCh. 17 - Prob. 17PCh. 17 - Prob. 18PCh. 17 - A 1.00-kg block of copper at 20.0C is dropped into...Ch. 17 - A resting adult of average size converts chemical...Ch. 17 - Prob. 21PCh. 17 - Prob. 22PCh. 17 - An ideal gas is enclosed in a cylinder with a...Ch. 17 - Prob. 24PCh. 17 - Prob. 25PCh. 17 - A sample of an ideal gas goes through the process...Ch. 17 - A thermodynamic system undergoes a process in...Ch. 17 - A gas is taken through the cyclic process...Ch. 17 - Consider the cyclic process depicted in Figure...Ch. 17 - Why is the following situation impossible? An...Ch. 17 - An ideal gas initially at 300 K undergoes an...Ch. 17 - In Figure P17.32, the change in internal energy of...Ch. 17 - Prob. 33PCh. 17 - Prob. 34PCh. 17 - Prob. 35PCh. 17 - Prob. 36PCh. 17 - Prob. 37PCh. 17 - One mole of an ideal gas does 3 000 J of work on...Ch. 17 - A 1.00-mol sample of hydrogen gas is heated at...Ch. 17 - A sample of a diatomic ideal gas has pressure P...Ch. 17 - Prob. 41PCh. 17 - Prob. 42PCh. 17 - Prob. 43PCh. 17 - Review. This problem is a continuation of Problem...Ch. 17 - Prob. 45PCh. 17 - A 2.00-mol sample of a diatomic ideal gas expands...Ch. 17 - Prob. 47PCh. 17 - An ideal gas with specific heat ratio confined to...Ch. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - Prob. 51PCh. 17 - Prob. 52PCh. 17 - Air (a diatomic ideal gas) at 27.0C and...Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - Prob. 59PCh. 17 - Prob. 60PCh. 17 - Prob. 61PCh. 17 - Prob. 62PCh. 17 - The surface of the Sun has a temperature of about...Ch. 17 - Prob. 64PCh. 17 - At high noon, the Sun delivers 1 000 W to each...Ch. 17 - A theoretical atmospheric lapse rate. Section 16.7...Ch. 17 - Prob. 67PCh. 17 - A sample of a monatomic ideal gas occupies 5.00 L...Ch. 17 - An aluminum rod 0.500 m in length and with a...Ch. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Prob. 75PCh. 17 - Prob. 76PCh. 17 - Prob. 77PCh. 17 - Prob. 78PCh. 17 - Prob. 79PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 84PCh. 17 - Prob. 85PCh. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Water in an electric teakettle is boiling. The...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A 200g copper bowl contains 100 g of wate, both at 25 degrees celsius. A very hot 300 g copper cylinder is dropped into the water. The final temperature of the system is 100 degrees celsius. Neglect energy transfer with the environment. The specific heat of water = 4186 J/kg. K and that of copper = 386J/kg. K. How much energy is transferred to the copper bowl as heat?arrow_forwardA 12 cm -diameter cylinder contains argon gas at 10 atm pressure and a temperature of 60 ∘C . A piston can slide in and out of the cylinder. The cylinder's initial length is 23 cm . 2600 J of heat are transferred to the gas, causing the gas to expand at constant pressure. What is the final temperature of the cylinder? What is the final length of the cylinder?arrow_forwardA 0.825 kg block of iron, with an average specific heat of 5.60 × 102 J/kg·K, is initially at a temperature of 254.0◦C. The block of iron is placed in a calorimeter with 16.4 g of water at 12.2◦C. What is the final thermal equilibrium temperature? If the answer if 100.0◦C, how much water is still in liquid form? Note: Treat the mass and heat capacity of the calorimeter as neglible.arrow_forward
- A 900 g copper rod at 20 degrees celcius has a length of 1.0000 m. The thermal expansion coefficient of copper is 17 x 10^-6 degrees celcius -1. The specific heat capacity is 0.385 kJ/kg degrees celcius. Question A: The copper is heated to 400 degrees celcius. What is the new length? Give the answer in meters and with 4 digits of precision after the decimal. Question B: The hot copper is then quenched by dunking the entire rod in a bucket with 10 kg of water at 20 degrees celcius. The specific heat capaciy of water is 4.18 kJ/kg degrees celcius. If none of the water turns to steam what is the equilibrium temp of the copper rod and water? Please give the answer in degrees celcius Question C: You measure the equilbrium temp and find that it is 24 degrees celcius. If the latent heat of vaporization of water is 2,260 kJ/kg, what mass of water turned to steam? Answer in gramsarrow_forwardWhile hanging out in Lab, you decide to conduct another calorimetry experiment, but this time, you want to do it on a bit larger scale. You place 3.8 kg of water in a large aluminum can that has a mass of 15 kg. You heat the water and can up to an initial temperature of 80◦ C, and then slowly add 400 g of ice that has an initial temperature of −10◦ C. You stir the ice and water until all of the ice melts, and the system comes to an equilibrium temperature of 32◦ C. You feel good about things until you realize that you did not cover the aluminum can and that some heat was lost to the environment during the experiment. Use the information provided to calculate the amount of heat that was lost to the environment.arrow_forwardWater is placed into an iron container. They are thermally isolated.The iron container has a mass of 600. g and is initially at 90.0 degrees Celsius. The water has a mass of 180. g and is initially at 30.0 degrees Celsius.What is the temperature of the container and water once they reach thermal equilibrium?arrow_forward
- When you apply the brakes on your car, the kinetic energy of your vehicle is transformed into thermal energy in your brake disks. During a mountain descent, a 28.00-cm-diameter iron brake disk heats up from 30°C to 180°C. What is the diameter of the disk after it heats up?arrow_forwardIn an electrically heated home, the temperature of the ground in contact with a concrete basement wall is 10.3 oC. The temperature at the inside surface of the wall is 18.1 oC. The wall is 0.14 m thick and has an area of 6.5 m2. Assume that one kilowatt hour of electrical energy costs $0.10. How many hours are required for one dollar's worth of energy to be conducted through the wall?arrow_forwardA solid sphere has a temperature of 827 K. The sphere is melted down and recast into a cube that has the same emissivity and emits the same radiant power as the sphere. What is the cube's temperature in kelvins? Number i T₂ Units Toarrow_forward
- Suppose that the rms speed of carbon dioxide molecules, with molar mass of 44.0 g/mol, in a flame is found to be 1.2 × 105 m/s a.What temperature, in kelvins, does this represent? b. What temperature, in celsius does this represent?arrow_forwardA classroom has dimensions 8.00 m x 10.00 m x 3.00 m. A 1000 W electric space heater is being used to warm the room from 5.00°C to 20.00°C on a cold morning. If the density of air is 1.29 kg/m°, and the specific heat capacity of air is 1004 J/(kg-K), how long will it take to heat the room? Assume no loss of thermal energy to the surroundings. A) 1.30 minutes B) 241 minutes C) 45.3 minutes O D) 77.7 minutesarrow_forwardJE JIE U 8. A 3.2 m length of copper pipe extends directly from a hot-water heater in a basement to a faucet on the first floor of a house. If the faucet isn't fixed in place, how much will it rise when the pipe is heated from 20.0° C to 85.9° C. The coefficient of linear expansion for copper is 1.6*10$ K-. An amergensy vahicla is traveling at 45 m/s annrnaching a car heading inarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON
Thermal Expansion and Contraction of Solids, Liquids and Gases; Author: Knowledge Platform;https://www.youtube.com/watch?v=9UtfegG4DU8;License: Standard YouTube License, CC-BY