
Concept explainers
a)
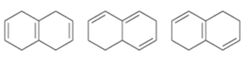
Interpretation:
The molecules given are to be arranged from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
Concept introduction:
Extended conjugation shifts the UV absorption maxima to higher wavelengths. For example, the UV absorption maxima for 1,3-butadiene is 217 nm and that of 1,3,5-hexatriene is 258nm.
To arrange:
The molecules given from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
b)
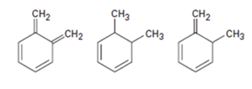
Interpretation:
The molecules given are to be arranged from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
Concept introduction:
Extended conjugation shifts the UV absorption maxima to higher wavelengths. For example, the UV absorption maxima for 1,3-butadiene is 217 nm and that of 1,3,5-hexatriene is 258nm.
To arrange:
The molecules given from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
c)
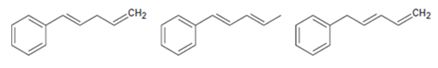
Interpretation:
The molecules given are to be arranged from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
Concept introduction:
Extended conjugation shifts the UV absorption maxima to higher wavelengths. For example, the UV absorption maxima for 1,3-butadiene is 217 nm and that of 1,3,5-hexatriene is 258nm.
To arrange:
The molecules given from shortest to longest wavelength on the basis their wavelength of maximum absorption in UV spectroscopy.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Ultraviolet and visible molecular absorption spectroscopy is used primarily for quantitative analysis. On the other hand Infrared absorption spectroscopy is a very powerful tool for determining the identity and structure of both inorganic and organic compounds. Which of these statements is correct? Only the first statement is correct Only the second statement is correct Both statements are correct None of the statements is correctarrow_forwardExplain in detail how the spectroscopic data shows that cyclohexanol, the reactant, produced the expected product, cyclohexene. Is the product pure? How do you know? Refer to specific frequencies in the IR spectrum and specific signals in the NMR spectra to support your conclusion.arrow_forwardWhat is the expected IR absorptions you would expect to see from the following molecule? OH If you were to take the IR of the starting molecule and then the product after the reaction is complete which IR absorption peaks will disappear and which will appear? Give absorption number and what part of the molecule that absorption references. H3O*arrow_forward
- Identify the type of organic compound, by NAME from their IR spectra givenarrow_forwardIR spectroscopy does not distinguish very well betweenisomers I and J. Explain why. How could UV–vis spectroscopy be used to distinguish between themarrow_forwardQUESTION 26 Arrange the following in order of INCREASING UV maximum wavelength of absorption (e. SHORTEST WAVELENGTH is #1-LONGEST WAVELENGTH is #4) -NH₂ OH -NH₂ -NH₂ 00 -NH3 -OH 00arrow_forward
- Based on this IR spectrum and 1H NMR spectrum and the given chemical formula is C9H10O2, what would be the chemical structure of the compound? Please give a full analysis of the NMR spectra for given compound.arrow_forwardshows five isomers of C5H10O2. The two structures that would be hardest to distinguish by IR spectroscopy arearrow_forwardHow would infrared spectroscopy be useful in distinguishing between the followingcompounds? Cite absorption bands you would expect from each.arrow_forward
- The IR spectrum of a certain compound is shown below. Given that this compound has a molecular formula of C5H8O, assign into detail the IR absorption peaks in the spectrum.arrow_forwardUsing Woodward–Fieser rules predict the following wavelength of the UV absorption band of the following(answer the encircled questions):arrow_forwardHow can IR spectroscopy be used to distinguish between each pair of isomers?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

