
Concept explainers
Interpretation:
The structure of a given molecular formula C8H16 to be predicted using spectrum details.
Concept introduction:
1HNMR : The 1HNMR spectrum gives information on the different electronic environment of protons. The number of signal (proton types) generated in 1HNMR are predicted by performing symmetry operations (rotation or reflection symmetry).
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
HDI Calculation:
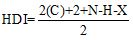
Where
C represent number of carbons.
N represent number of nitrogens.
H represent number of hydrogens.
X represent number of halogens.
To identify:
The structure of a given molecular formula C8H16.
Trending nowThis is a popular solution!

Chapter 13 Solutions
Organic Chemistry
- Saturated aldehyde Aromatic aldehyde At what position would you expect to observe IR absorption for the carbonyl groups in the following molecules? Infrared Absorptions of Some Aldehydes and Ketones. Carbonyl type Absorption cm-1 1730 1705 Example CH3CHO PhCHO a,ẞ-Unsaturated H2C=CHCHO 1705 aldehyde Saturated ketone CH3 COCH3 1715 Cyclohexanone 1715 Cyclopentanone 1750 Cyclobutanone Aromatic ketone 1785 PhCOCH3 a,ẞ-Unsaturated ketone H2C=CHCOCH3 1685 1690 Molecule #1: Molecule #2: CHO CHO CH3 cm-1 cm -1arrow_forwardHow many signals are expected in the decoupled ¹³C NMR spectrum of 3-bromotoluene? Br CH3arrow_forwardThe mass spectrum of 1-ethyl-1-methylcyclohexane shows many fragments, with two in very large abundance. Kne appears af m/z=111 and the other appears at m/z=97. Identify the structure of each of these fragments.arrow_forward
- How many different 13C-absorption lines and how many 'H- resonances (disregard splitting and assume that solvent exchange of acidic hydrogens does NOT take place) are observed in the spectrum of each of the following compounds? (H3C)3C-C(CH3)2 CI A H3C (H3C)3C B CH3 CH3 H₂C= -COOH C CH-CI H3C-C-CH Cl Br Darrow_forwardWhich one of these four compounds would give a 13C NMR spectrum with only these three peaks, one at 210, one at 40 and one at 20? H3C OB OD 13C NMR delta (8) values C=O of ketones, aldehydes C=O of acids, esters, amides C=C and C=N C-X (where X = halogen, O) base position, alkyl C OC A OA CH3 H3C B CH3 H3C1 220-200 160-180 100-150 40-70 0 - 40 CH3 H₂C D CH3arrow_forwardWith 1H-NMR, how many signals/peaks would you expect for 1-chloro-3-methylbutane?arrow_forward
- What is the structure from the formula C10H12O and the spectra?arrow_forward10- The strongest absorption area of the benzene ring in visible and ultraviolet spectroscopy at 255 nm 11- A Calverti rearrangement occurs when the compound has a hydrogen atom in theẞ. Position 12-Carboxylic acids show an absorption peak of 44 m/e Q/ true or falsearrow_forwardIdentify the significant absorption peaks by labeling them right on the spectrumand includethe spectrum in your laboratory report. Absorption peaks corresponding to the followinggroups should be identified: C—H (SP3) C—H (SP2) C—H (aldehyde) O—H C=O C=C (aromatic)aromatic substitution pattern C—OC—X (if applicable)arrow_forward
- Select the partial structure corresponding to a peak at 170 ppm in the ¹3C spectrum. OH-C=O OO-C=O OC-C=C OC-C=Oarrow_forwardThe 1H-NMR spectrum of 1,3-propanediol (HO-CH2-CH2-CH2-OH) shows a quintet at 1.81 ppm, a singlet at 2.75 pm, and a triplet at 3.83 ppm. Assign each signal to the protons it corresponds to in the molecule. Explain the splitting pattern observed for each signal.arrow_forwardA'2 The 'H-NMR spectra of cyclohexanol and cyclohexanone are given below. Identify which spectrum belongs to which compound and assign the peaks in each spectrum that substantiate your decision.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
