
Concept explainers
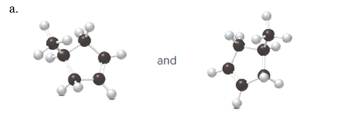
(a)
Interpretation:
Each pair from the following structures should be identified as constitutional isomers, stereo isomers or identical.
Concept Introduction:
Isomers are the different molecules which have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
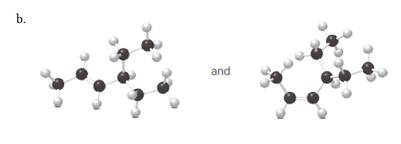
(b)
Interpretation:
Each pair from the following structures should be identified as constitutional isomers, stereo isomers or identical.
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Isomers are the different molecules which have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Cyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forwardIs the compound CH3CH=CH2 saturated or unsaturated? Explain.arrow_forwardAre compounds with different placement of double/triple bonds considered stereoisomers or constitutional isomers?arrow_forward
- 8. Circle and clearly label five functional groups. 'N' `N' LOH Tubulysin D IZarrow_forwardSub Question 27 of 31 The molecules shown below are A) Identical molecules B) Stereoisomers C) Constitutional isomers D) Isotopes E) Neither identical nor a type of isomerarrow_forwardWhy are these two structures enantiomers? They look more like structural isomers to me.arrow_forward
- 1- What are the functional groups in diethylstilbesterol ? 2-How many Chiral carbon atom are in diethylstilbesterol? + identify themarrow_forwarddraw structure for 4-bromo-2-pentene where the double bond has a Z geometry and the chiral carbon an S configuration ?arrow_forwardwhat are the stereoisomers of propane and chloropropane.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





